Rutile and CaCl2 Structure of SnO2 Phase Transition under High-Pressure Studied by First-Principles Method
-
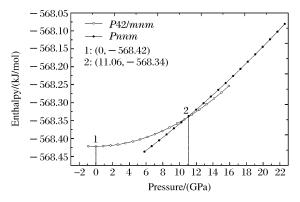
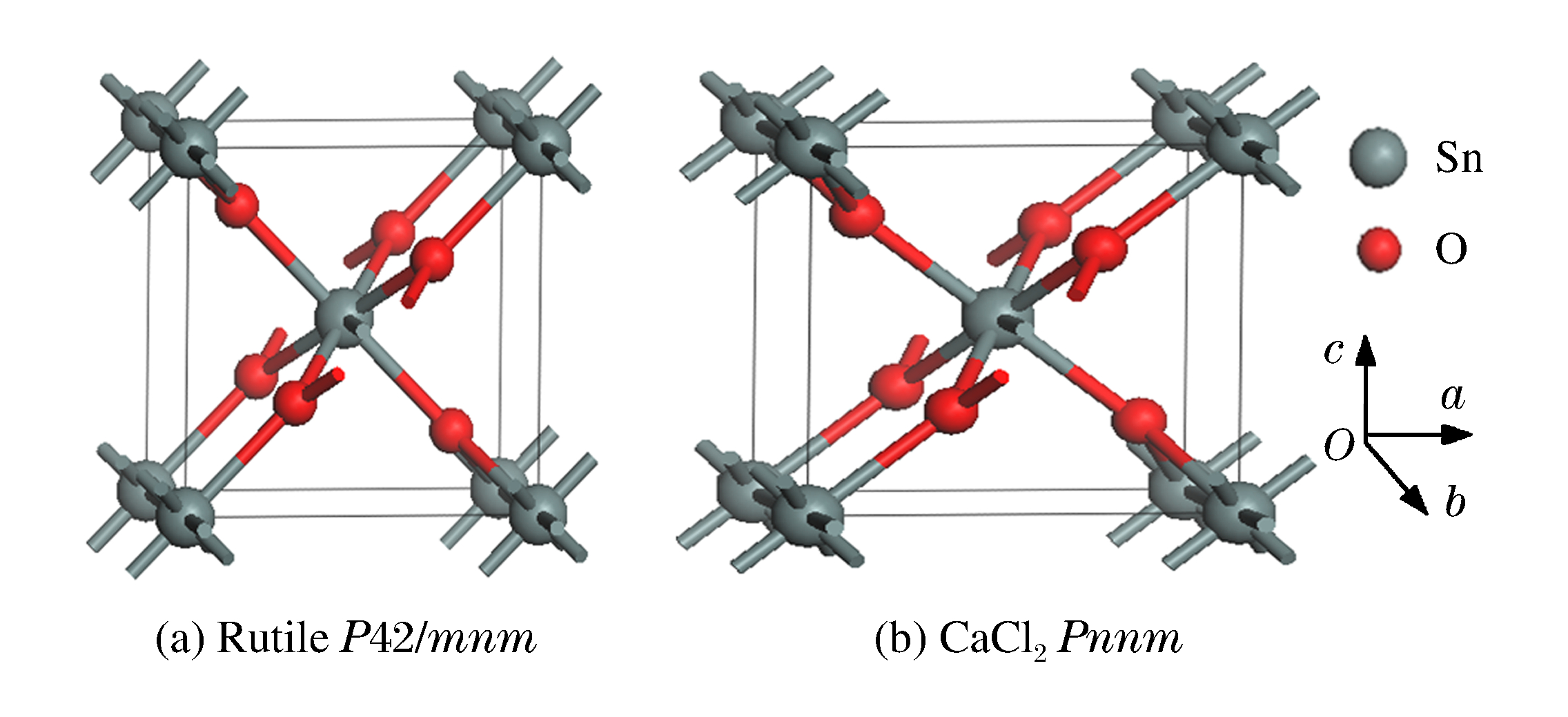
摘要: 采用基于密度泛函理论(DFT)的平面波超软赝势法,用广义梯度近似(GGA)PBE交换相关泛函,对P42/mnm和Pnnm晶体结构SnO2的力学性质进行第一性原理计算。计算表明,两种结构SnO2的晶体结构参数与实验和理论值符合较好。随着压强的增大,P42/mnm型SnO2的生成焓呈抛物线状缓慢增大,Pnnm型SnO2的生成焓为一直线迅速上升。P42/mnm结构弹性常数随压强增加规律增大,Pnnm结构弹性常数无规律性变化,外界压强为11.06 GPa时发生相变。两结构中低指数面中Pnnm结构的{100}晶面族之间滑移的可能性最大、最不稳定,{001}晶面族间结合最牢固。金红石型SnO2维氏硬度计算理论值为10.49 GPa,Pnnm结构为11.42 GPa。德拜温度计算表明,结构对称性高低的差异,造成P42/mnm结构德拜温度在一个小范围内波动,而Pnnm结构德拜温度在8~10 GPa内有突变,呈缓慢下降的趋势。Abstract: In the present work we performed the first principles calculations based on the density functional theory and investigated the P42/mnm-Pnnm phase transition and the mechanical properties of SnO2.For the exchange-correction energy we employed the generalized gradient approximation (GGA) in the Perdew, Burke and Ernzerhof (PBE) form.The elastic stiffness coefficients, cij, bulk modulus, shear modulus, Young modulus, Reuss modulus, Voigt modulus and anisotropy factor were calculated for two polymorphs of SnO2:rutile and CaCl2 structure.Our results for the structural parameters and elastic constants at the equilibrium phase are in good agreement with the available theoretical and experimental values.Using the enthalpy-pressure data, we observed the rutile to CaCl2 structural phase transition under 11.06 GPa pressure.In the low index plane of two polymorphs' SnO2, the most unstable and the possibility of slippage crystal planes was the {100} crystal planes of Pnnm structure, and the {001} crystal planes has the strongest binding force.The Vickers hardness calculated for the P42/mnm of SnO2 was 10.49 GPa, and the Pnnm structure was 11.42 GPa.Because of the differences in the level of the structure symmetry, results from calculation show that the Debye temperature waverd in a small range for the utile structure but it had mutations in 8-10 GPa and showed a downward trend for the CaCl2 structure.
-
Key words:
- tin oxide /
- pressure /
- first principles /
- phase transition /
- elastic stiffness coefficients /
- Debye temperature
-
表 1 几何优化后不同SnO2晶体结构的晶胞参数
Table 1. Lattice constants of different SnO2 crystal structures
Space p/(GPa) Unit-cell parameter/(nm) Volume/(nm3) Space p/(GPa) Unit-cell parameter/(nm) Volume/(nm3) a b c a' b' c' P42/mnm 0 0.468 64 0.468 64 0.315 57 0.069 31 Pnnm 6 0.467 76 0.460 02 0.314 02 0.067 57 2 0.467 02 0.467 02 0.315 06 0.068 72 8 0.467 29 0.457 97 0.313 46 0.067 08 4 0.465 44 0.465 44 0.314 61 0.068 16 10 0.466 32 0.454 84 0.312 63 0.066 31 6 0.464 09 0.464 09 0.313 97 0.067 62 12 0.465 49 0.452 87 0.312 27 0.065 83 8 0.462 55 0.462 55 0.313 69 0.066 99 14 0.464 10 0.451 71 0.311 85 0.065 38 10 0.461 42 0.461 42 0.313 09 0.066 66 16 0.463 70 0.450 06 0.311 27 0.064 96 12 0.460 14 0.460 14 0.312 72 0.066 21 18 0.461 92 0.449 14 0.311 06 0.064 53 14 0.458 66 0.458 66 0.312 58 0.065 76 20 0.462 17 0.446 45 0.310 48 0.064 06 表 2 各向异性系数及各向异性压缩因子理论计算值
Table 2. Calculated shear anisotropic factors A1, A2, A3, and AB, AG for SnO2
Species A1 A2 A3 AB/(%) AG/(%) P42/mnm 1.030 1 1.030 1 5.969 3 1.42 16.33 Pnnm 0.955 1 1.038 6 8.227 6 7.29 25.84 表 3 两不同结构维氏硬度
Table 3. Calculated Vickers hardness of SnO2 type of P42/mnm and Pnnm
Species Coordination ZSn ZO nSn nO d/(nm) Ne/(nm-3) HVx/(GPa) HV/(GPa) P42/mnm Pol(1) 5 6 4 2 0.199 0 478 11.83 10.49 Pol(2) 4 6 4 2 0.201 0 418 10.08 13.80[16] Pnnm Pol(1) 5 6 4 2 0.197 8 310 12.73 11.42 Pol(2) 4 6 4 2 0.198 5 400 11.02 表 4 两不同结构的横波、纵波、总晶格振动波矢和德拜温度
Table 4. Transverse, longitudinal, average elastic wave velocities and Debye temperature for P42/mnm and Pnnm structures
Space p/(GPa) νP νS νm ΘD/(K) P42/mnm 0 3.95 7.29 5.19 492 2 4.02 7.42 5.29 508 4 4.03 7.50 5.30 507 6 3.91 7.52 5.19 494 8 4.03 7.68 5.34 509 10 3.92 7.67 5.21 495 12 3.96 7.78 5.29 501 14 3.89 7.78 5.21 492 Pnnm 6 3.02 7.68 5.12 488 8 3.00 7.04 5.23 497 10 2.96 6.31 4.12 390 12 2.94 7.11 4.06 384 14 2.92 7.14 4.04 381 16 2.90 7.54 4.03 378 18 2.89 7.27 4.02 375 20 2.86 6.99 3.97 371 -
[1] FU H Z, LIU W F, PENG F, et al.Phase transition and thermodynamic properties of MgTe under high pressure [J].J Alloy Compd, 2009, 480(2):587-591. doi: 10.1016/j.jallcom.2009.02.009 [2] 蒋华龙, 周大伟, 刘旭焱, 等.压力下MoS2的结构相变以及热动力学性质的第一原理研究[J].低温物理学报, 2014, 36(5):379-386. http://d.g.wanfangdata.com.cn/Thesis_Y2614813.aspxJIANG H L, ZHOU D W, LIU X Y, et al.First-principles investigation on the structural transition and thermodynamic properties of MoS2 under pressure [J].Chinese Journal of Low Temperature Physics, 2014, 36(5):379-386. http://d.g.wanfangdata.com.cn/Thesis_Y2614813.aspx [3] 黄晓玉, 何朝政, 宋海珍, 等.分子动力学方法模拟Be高温和高压的状态方程和力学性质[J].西南民族大学学报(自然科学版), 2014, 40(6):921-927. http://d.old.wanfangdata.com.cn/Periodical/xnmzxyxb201406021HUANG X Y, HE C Z, SONG H Z, et al.Study of equations of state and mechanical property of Be by molecular dynamics method [J].Journal of Southwest University for Nationalities(Natural Science Edition), 2014, 40(6):921-927. http://d.old.wanfangdata.com.cn/Periodical/xnmzxyxb201406021 [4] KUANG F G, KUANG X Y, KANG S Y, et al.A first principle study of pressure-induced effects on phase transitions, band structures and elasticity of zinc oxide [J].Mater Sci Semi Process, 2014, 23:63-71. doi: 10.1016/j.mssp.2014.02.029 [5] ALPTEKIN S, DURANDURDU M.Pressure-induced phase transition of BeO [J].Solid State Commun, 2009, 149(9):345-348. http://www.sciencedirect.com/science/article/pii/S0038109808007357 [6] BAI L, LI Q, CORR S A, et al.Pressure-induced phase transitions and metallization in VO2 [J].Phys Rev B, 2015, 91(10):104110. doi: 10.1103/PhysRevB.91.104110 [7] CUI S, FENG W, HU H, et al.Pressure-induced structural transition in PuTe [J].Phys Lett A, 2009, 373(18):1693-1696. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=7630c2f0f2e7b8a203e9beee5d0bc012 [8] 张旭东, 史海峰, 权善玉.ZnS和MgS的高压相变和弹性性质[J].沈阳工业大学学报, 2011, 33(1):50-54. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK201100149739ZHANG X D, SHI H F, QUAN S Y.Phase transition and elastic properties of ZnS and MgS under high pressure [J].Journal of Shenyang University of Technology, 2011, 33(1):50-54. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK201100149739 [9] BILGE M, ÖZDEMIR KART S, KART H H, et al.B3—B1 phase transition and pressure dependence of elastic properties of ZnS [J].Mater Chem Phys, 2008, 111(2/3):559-564. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=3c934556cb8f8c1cad37af6e1a1da854 [10] SHIEH S R, KUBO A, DUFFY T S, et al.High-pressure phases in SnO2 to 117 GPa [J].Phys Rev B, 2006, 73(1):014105. doi: 10.1103/PhysRevB.73.014105 [11] HAINES J, LÉGER J M.X-ray diffraction study of the phase transitions and structural evolution of tin dioxide at high pressure:relationships between structure types and implications for other rutile-type dioxides [J].Phys Rev B, 1997, 55(17):11144. doi: 10.1103/PhysRevB.55.11144 [12] LEVY M.Universal variational functionals of electron densities, first-order density matrices, and natural spin-orbitals and solution of the v-representability problem [J].Proc Nat Acad Sci, 1979, 76(12):6062-6065. doi: 10.1073/pnas.76.12.6062 [13] PERDEW J P, BURKE K, ERNZERHOF M.Generalized gradient approximation made simple [J].Phys Rev Lett, 1996, 77(18):3865. doi: 10.1103/PhysRevLett.77.3865 [14] HOHENBERG P, KOHN W.Inhomogeneous electron gas [J].Phys Rev, 1964, 136(3B):B864. doi: 10.1103/PhysRev.136.B864 [15] KRESSE G, FURTHMVLLER J.Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set [J].Phys Rev B, 1996, 54(16):11169. doi: 10.1103/PhysRevB.54.11169 [16] SEKI H.High-temperature structures of the rutile-type oxides, TiO2 and SnO2 [J].J Ceram Soc Jap, 1984, 92(4):219. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=J-STAGE_2284613 [17] 冯晶, 陈敬超, 肖冰, 等.Ag-Sn合金的氧化过程与热力学性质[J].物理化学学报, 2008, 24(11):2007-2012. doi: 10.3866/PKU.WHXB20081112FENG J, CHEN J C, XIAO B, et al.Oxidation and thermodynamic properties of Ag-Sn alloy [J].Acta Phys Chim Sin, 2008, 24(11):2007-2012. doi: 10.3866/PKU.WHXB20081112 [18] THEOCARIS P S, SOKOLIS D P.Invariant elastic constants and eigentensors of orthorhombic, tetragonal, hexagonal and cubic crystalline media [J].Acta Crystall Sec A:Found Crystall, 2000, 56(4):319-331. doi: 10.1107/S0108767300001926 [19] SCHREIBER E, ANDERSON O L, SOGA N, et al.Elastic constants and their measurement [M].New York:McGraw-Hill, 1974. [20] RAVINDRAN P, FAST L, KORZHAVYI P A, et al.Density functional theory for calculation of elastic properties of orthorhombic crystals:application to TiSi2 [J].J Appl Phys, 1998, 84(9):4891-4904. doi: 10.1063/1.368733 [21] HUANG H J, XU J.First-principles study into the effect of substitutional Al alloying on the mechanical properties and electronic structure of D88-Ti5Si3 [J].Acta Phys Chim Sin, 2015, 31(2):253-260. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=wlhxxb201502008 [22] CHUNG D H, BUESSEM W R.The elastic anisotropy of crystals [J].J Appl Phys, 1967, 38(5):2010-2012. doi: 10.1063/1.1709819 [23] GAO F, HE J, WU E, et al.Hardness of covalent crystals [J].Phys Rev Lett, 2003, 91(1):015502. doi: 10.1103/PhysRevLett.91.015502 [24] HE J L, GUO L C, YU D L, et al.Hardness of cubic spinel Si3N4 [J].Appl Phys Lett, 2004, 85(23):5571. doi: 10.1063/1.1832756 [25] 张建新, 王海燕, 高爱华, 等.Mg-Sn-Si系合金的热力学基础及合金相演变过程分析[J].物理学报, 2015, 64(6):066401. http://d.old.wanfangdata.com.cn/Periodical/wlxb201506034ZHANG J X, WANG H Y, GAO A H, et al.Study on thermodynamics basic and alloy phase evolution of Mg-Sn-Si magnesium alloy [J].Physics, 2015, 64(6):066401. http://d.old.wanfangdata.com.cn/Periodical/wlxb201506034 [26] 黄昆, 韩汝琦.固体物理学[M].北京:高等教育出版社, 2002:126-132.HUANG K, HAN R Q.Solid state physics [M].Beijing:Higher Education Press, 2002:126-132. -







 下载:
下载: