Preparation and Pressure-Induced Cubic-to-Orthorhombic Phase Transition in ZrW2O8
-
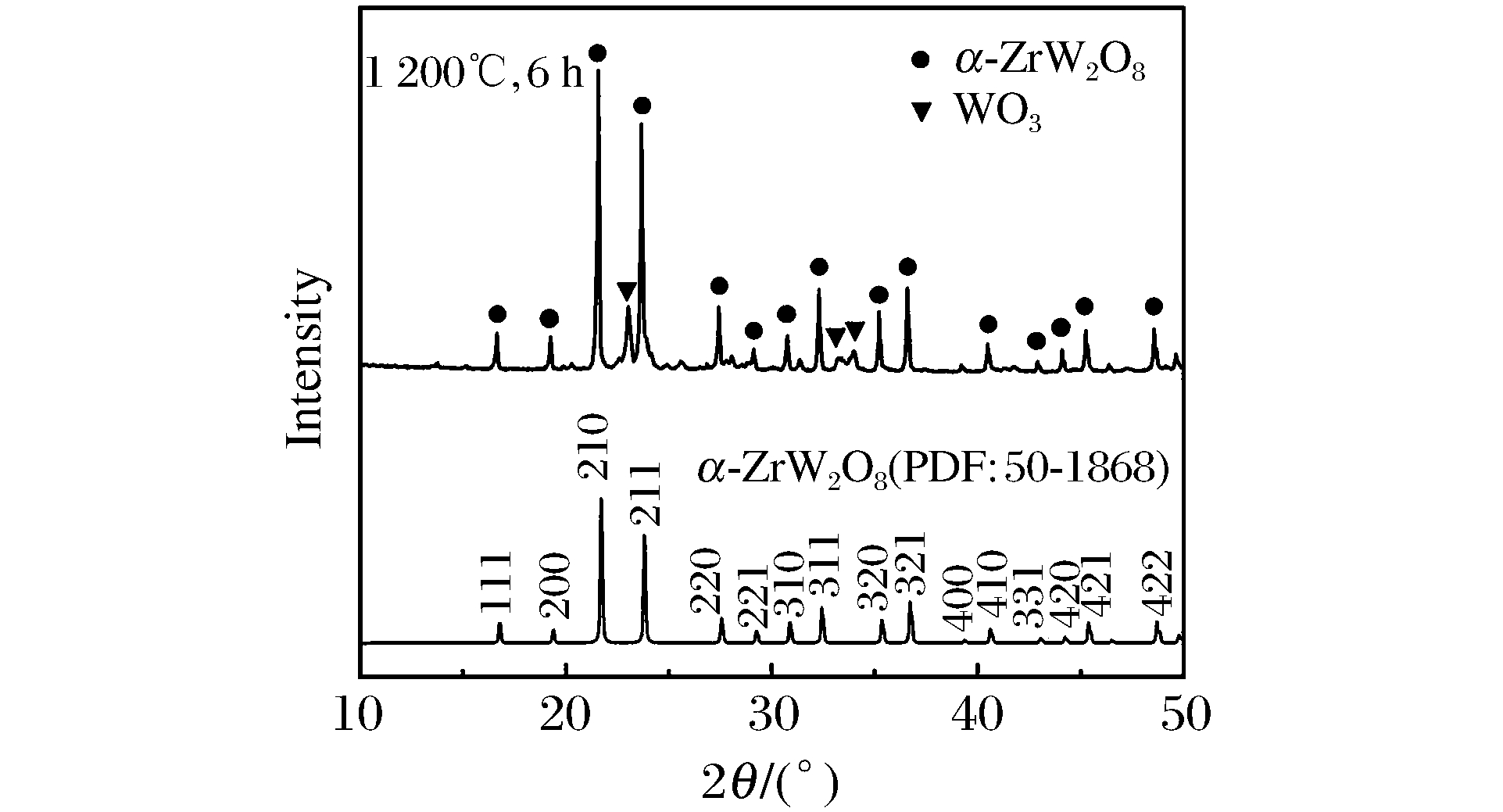
摘要: 采用固相烧结法制备了纯度较高的钨酸锆(ZrW2O8)粉体,并利用金刚石对顶压砧(DAC)对其进行了原位高压同步辐射X射线衍射研究。结果表明:在常温下,压力0.27 GPa附近, 晶体的对称性降低, 并发生α-ZrW2O8向γ-ZrW2O8晶体结构相变; 压力3.81 GPa附近,γ-ZrW2O8完全转变为非晶相。通过拟合得到α-ZrW2O8的体弹模量B=(40.0±3.8)GPa,γ-ZrW2O8的体弹模量B=(37.0±3.0)GPa。
-
关键词:
- 钨酸锆(ZrW2O8) /
- 负热膨胀 /
- 高压相变 /
- 同步辐射 /
- 体弹模量
Abstract: Negative thermal expansion (NTE) ZrW2O8 powders were prepared by solid-state reaction with ZrO2 and WO3 powders.In situ high-pressure X-ray diffraction measurements for powder ZrW2O8 were performed by using a diamond anvil cell (DAC) with synchrotron radiation facility.The result shows that powder ZrW2O8 is cubic phase with a little WO3.At about 0.27 GPa and room temperature, theα-ZrW2O8 phase transforms into a quenchable orthorhombic structure called the γ phase.Upon further compression, the γ phase becomes amorphous at the pressure of 3.81 GPa.The obtained bulk modulus of α-ZrW2O8 and γ-ZrW2O8 are (40.0±3.8)GPa and (37.0±3.0)GPa. -
-
[1] Mary T A, Evans J S O, Vogt T, et al. Negative thermal expansion from 0.3 to 1 050 Kelvin in ZrW2O8[J]. Science, 1996, 272(5258): 90-92. doi: 10.1126/science.272.5258.90 [2] Sun Z H, Song X Y, Yin F X, et al. Giant negative thermal expansion in ultrafine-grained Mn3(Cu1-xGex)N(x=0.5)bulk[J]. J Phys D: Appl Phys, 2009, 42(12): 122004. doi: 10.1088/0022-3727/42/12/122004 [3] Mary T A, Evans J S O, Sleight A W, et al. Negative thermal expansion in Sc2(WO4)3[J]. Solid State Chem, 1998, 137(1): 148-160. doi: 10.1006/jssc.1998.7744 [4] Evans J S O, Mary T A, Vogt T, et al. Negative Thermal expansion in ZrW2O8 and HfW2O8[J]. Chem Mater, 1996, 8(12): 2809-2823. doi: 10.1021/cm9602959 [5] Kowach G R. Growth of single crystals of ZrW2O8[J]. J Crystal Growth, 2000, 212(1/2): 167-172. http://www.sciencedirect.com/science/article/pii/S0022024899008921 [6] Isobe T, Kato Y, Mizutani M, et al. Pressureless sintering of negative thermal expansion ZrW2O8/Zr2WP2O12 composites[J]. Mater Lett, 2008, 62(23): 3913-3915. doi: 10.1016/j.matlet.2008.05.046 [7] Forster P M, Sleight A W. Negative thermal expansion in Y2W3O8[J]. Int J Inorgan Mater, 1999, 1(2): 123-127. doi: 10.1016/S1466-6049(99)00021-5 [8] Evans J S O, Hu Z, Jorgensen J D, et al. Compressibility, phase transitions, and oxygen migration in zirconium tungstate, ZrW2O8[J]. Science, 1997, 275(5296): 61-65. doi: 10.1126/science.275.5296.61 [9] Mittal R, Chaplot S L. Lattice dynamical calculation of isotropic negative thermal expansion in ZrW2O8 over 0-1 050 K[J]. Phys Rev B, 1999, 60(10): 7234-7237. doi: 10.1103/PhysRevB.60.7234 [10] Jorgensen J D, Hu Z, Teslic S, et al. Pressure-induced cubic-to-orthorhombic phase transition in ZrW2O8[J]. Phys Rev B, 1999, 59(1): 215-225. doi: 10.1103/PhysRevB.59.215 [11] Perottoni C A, da Jornada J A H. Pressure-induced amorphization and negative thermal expansion in ZrW2O8[J]. Science, 1998, 280(5365): 886-889. doi: 10.1126/science.280.5365.886 [12] Pantea C, Migliori A, Littlewood P B, et al. Pressure-induced elastic softening of monocrystalline zirconium tungstate at 300 K[J]. Phys Rev B, 2006, 73(21): 214118. doi: 10.1103/PhysRevB.73.214118 [13] Drymiotis F R, Ledbetter H, Betts J B, et al. Monocrystal elastic constants of the negative-thermal-expansion compound zirconium tungstate(ZrW2O8)[J]. Phys Rev Lett, 2004, 93(2): 025502. doi: 10.1103/PhysRevLett.93.025502 [14] Liu W, Li B S. Elasticity of amorphous zirconium tungstate at high pressure[J]. Appl Phys Lett, 2008, 93(19): 191904. doi: 10.1063/1.3023049 -







 下载:
下载: