Equation of State of Detonation Products for RDX Explosive
-
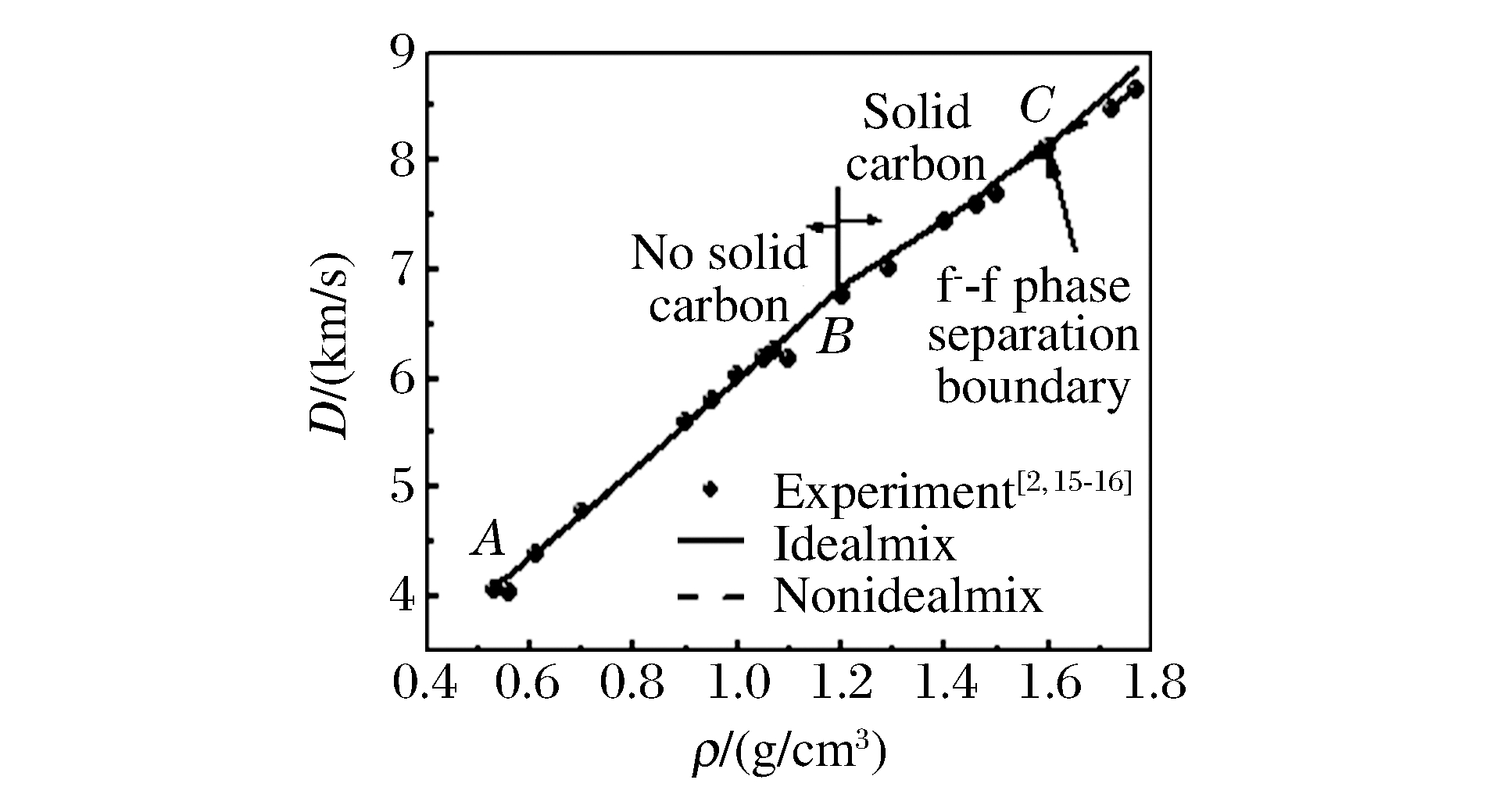
摘要: 采用基于统计物理的爆轰产物物态方程模型, 研究了不同初始密度下RDX炸药爆轰产物性质,发现炸药初始密度小于1.20 g/cm3时,炸药爆轰后只有气相产物生成,随着炸药初始密度进一步增大,爆轰后有固相碳析出。炸药初始密度小于1.60 g/cm3时,各气相产物按均匀混合处理,计算的爆速、爆压和实验值符合较好。随着炸药初始密度进一步增大,计算的爆速、爆压与实验值出现较大的偏差,经分析发现此处可能出现了超临界流体相分离现象,在物态方程模型中考虑了这种效应后,得到较好的结果。Abstract: The equation of state of detonation products for RDX explosive at different initial densities is calculated by adopting the program based on the statistical physics model.It is found that only gas phase product generates after the explosive detonation when the initial density of explosive is less than 1.20 g/cm3.With the increasing of initial density of explosive, solid carbon dissolves out after the explosive detonation.When the initial density of explosive is less than 1.60 g/cm3, the calculated detonation velocity and detonation pressure is in good consistent with the experimental value regarding the gas phase product as uniform mixing.With the increasing of initial density of explosive, the calculated detonation velocity and detonation pressure deviate largely from the experimental value.After detailed analysis, we find out that there is a supercritical fluid phase separation in this condition.Considering this effect in our program, a good result is obtained.
-
表 1 不同初始密度下RDX炸药CJ点的爆速、爆压的比较
Table 1. Detonation velocity and detonation pressure of RDX explosive of different initial density at CJ point
Initial density
/(g/cm3)DCJ/(km/s) pCJ(GPa) Experiment Idealmix Nonidealmix Experiment This work 0.53 4.06 4.10 - 2.90 0.56 4.05 4.21 3.16 3.19 0.61 4.39 4.40 - 3.73 0.70 4.78 4.75 4.72 4.86 0.90 5.61 5.58 - 8.06 0.95 5.80 5.79 9.46 9.02 1.00 6.04 6.00 - 10.03 1.05 6.19 6.21 - 11.11 1.07 6.26 6.30 11.6 11.56 1.10 6.18 6.42 12.0 12.25 1.20 6.75 6.85 15.2 14.71 1.29 7.00 7.13 16.4 17.14 1.40 7.44 7.46 21.3 20.37 1.46 7.60 7.66 20.8 22.26 1.50 7.69 7.82 7.81 - 23.60 1.59 8.10 8.19 8.14 28.7 28.66 1.60 8.13 8.25 8.18 26.0 27.01 1.72 8.46 8.65 8.48 30.8 31.46 1.77 8.64 8.89 8.70 33.8 32.82 -
[1] Chirat R, Pittion-Rossillon G. A new equation of state for detonation products[J]. J Chem Phys, 1981, 74(8): 4634-4642. doi: 10.1063/1.441653 [2] Mader C L. Numerical Modeling of Detonation[M]. Berkeley, CA: University of California Press, 1979. [3] 龙新平, 何碧, 蒋小华, 等.论VLW状态方程[J].高压物理学报, 2003, 17(4): 247-254.Long X P, He B, Jiang X H, et al. Discussions on the VLW equation of state[J]. Chinese Journal of High Pressure Physics, 2003, 17(4): 247-254. (in Chinese) [4] Ree F H. A statistical mechanical theory of chemically reacting multiphase mixtures: Application to the detonation properties of PETN[J]. J Chem Phys, 1984, 81(3): 1251-1263. doi: 10.1063/1.447811 [5] Lu J P. Evaluation of the thermochemical code—CHEETAH 2.0 for modeling explosives performance, DSTO-TR-1199[R]. 2001. [6] 赵艳红, 刘海风, 张弓木.基于统计物理的爆轰产物物态方程研究[J].物理学报, 2007, 56(8): 4791-4797. http://www.cnki.com.cn/Article/CJFDTotal-WLXB200708072.htmZhao Y H, Liu H F, Zhang G M. Equation of state of detonation products based on statistical mechanical theory[J]. Acta Phys Sin, 2007, 56(8): 4791-4797. (in Chinese) http://www.cnki.com.cn/Article/CJFDTotal-WLXB200708072.htm [7] 赵艳红, 刘海风, 张弓木, 等.高温高压下爆轰产物分子间相互作用的研究[J].物理学报, 2011, 60(12): 123401. http://www.cnki.com.cn/Article/CJFDTotal-WLXB201112025.htmZhao Y H, Liu H F, Zhang G M, et al. Pair interactions of detonation products at high pressure and high temperature[J]. Acta Phys Sin, 2011, 60(12): 123401. (in Chinese) http://www.cnki.com.cn/Article/CJFDTotal-WLXB201112025.htm [8] 赵艳红, 刘海风, 张其黎.高温高压下爆轰产物中不同种分子间的相互作用[J].物理学报, 2012, 61(23): 230509. http://d.wanfangdata.com.cn/Periodical/wlxb201223018Zhao Y H, Liu H F, Zhang Q L. Unlike-pair interactions of detonation products at high pressure and high temperature[J]. Acta Phys Sin, 2012, 61(23): 230509. (in Chinese) http://d.wanfangdata.com.cn/Periodical/wlxb201223018 [9] 董贺飞, 赵艳红, 洪滔. HMX炸药燃烧转爆轰数值模拟[J].高压物理学报, 2012, 26(6): 601-607. http://www.cnki.com.cn/Article/CJFDTotal-GYWL201206000.htmDong H F, Zhao Y H, Hong T. Numerical simulation of the deflagration-to-detonation transition behavior of explosive HMX[J]. Chinese Journal of High Pressure Physics, 2012, 26(6): 601-607. (in Chinese) http://www.cnki.com.cn/Article/CJFDTotal-GYWL201206000.htm [10] Fried L E, Howard W M. Explicit Gibbs free energy equation of state applied to the carbon phase diagram[J]. Phys Rev B, 2000, 61(13): 8734-8743. doi: 10.1103/PhysRevB.61.8734 [11] 刘福生, 陈先猛, 陈攀森, 等.液态CO2高温高密度状态方程研究[J].高压物理学报, 1998, 12(1): 28-33. http://www.cnki.com.cn/Article/CJFDTotal-GYWL801.004.htmLiu F S, Chen X M, Chen P S, et al. Equation of sate of liquid CO2 at high temperatures ang high densities[J]. Chinese Journal of High Pressure Physics, 1998, 12(1): 28-33. (in Chinese) http://www.cnki.com.cn/Article/CJFDTotal-GYWL801.004.htm [12] 杨向东, 胡栋, 经福谦.炸药爆轰产物液氮、液氦和水状态方程研究[J].高压物理学报, 1999, 13(2): 93-102. http://d.wanfangdata.com.cn/Periodical/gywlxb199902003Yang X D, Hu D, Jing F Q. Studies of EOS for detonation products: Liquid nitrogen, liquid helium and water[J]. Chinese Journal of High Pressure Physics, 1999, 13(2): 93-102. (in Chinese) http://d.wanfangdata.com.cn/Periodical/gywlxb199902003 [13] Ross M, Ree F H. Repulsive force of simple molecules and mixtures at high density and temperature[J]. J Chem Phys, 1980, 73(12): 6146-6152. doi: 10.1063/1.440106 [14] Ree F H. Simple mixing rule for mixtures with exp-6 interactions[J]. J Chem Phys, 1983, 78(1): 409-415. doi: 10.1063/1.444517 [15] Ree F H. Supercritical fluid phase separations: Implications for detonation properties of condensed explosives[J]. J Chem Phys, 1986, 84(10): 5845-5856. doi: 10.1063/1.449895 [16] Souers P C. Detonation equation of state at LLNL, UCRL-ID-119262[R]. Livermore, CA: Lawrence Livermore National Laboratory, 1995. -







 下载:
下载: