Shock Synthesis of a New B-C-N Compound
-
摘要: 理论预言,B-C-N化合物可能存在多种结构,并具有优良的物理性能,因此其合成研究引起了各国学者的关注。运用二级轻气炮冲击加载与样品回收技术,在40 GPa压力附近探索了B-C-N化合物的新相合成条件。X射线衍射、高分辨率透射电镜、电子衍射、X射线光电子能谱等检测技术的分析结果表明,3种不同原子比例的B-C-N前驱物在40 GPa的强冲击合成条件下,均能生成一种新的B-C-N致密新相单晶体,尺寸约为60 nm。具有这种结构的B-C-N化合物在文献中未见报道。研究结果对进一步探索B-C-N新相的结构形态及其高温高压合成条件具有参考意义,也为理论工作者提供了一种新的B-C-N结构模型。Abstract: Theory predicts that B-C-N compounds may exist in many structural forms and have excellent physical properties.Synthesis of such materials is attracting the attentions of scientists.A two-stage light-gas gun and the corresponding shock-recovery technology were applied to explore the synthesis conditions of B-C-N compound of new phase structures under 40 GPa pressure.XRD (X-ray diffraction), HRTEM (high-resolution transmission electron microscopy), ED (electron diffraction) and XPS (X-ray photoelectron spectroscopy) analysis show that all three precursors with different atomic ratio of B, C and N can generate a new dense phase of B-C-N compound under strong shock loading of 40 GPa.Single crystals of 60 nm are directly observed in the recovery sample.The new B-C-N compound of such structure has not been reported in the literature before.Therefore, this discovery is of significance for exploring new structures of B-C-N compounds and the synthesis conditions in future; meanwhile, a new structure of B-C-N compound is waiting to be studied by theoretical researchers.
-
Key words:
- B-C-N compounds /
- superhard material /
- shock wave synthesis /
- light gas gun
-
表 1 B-C-N冲击合成的前驱物
Table 1. Pyrolysis precursor for shock synthesis of B-C-N
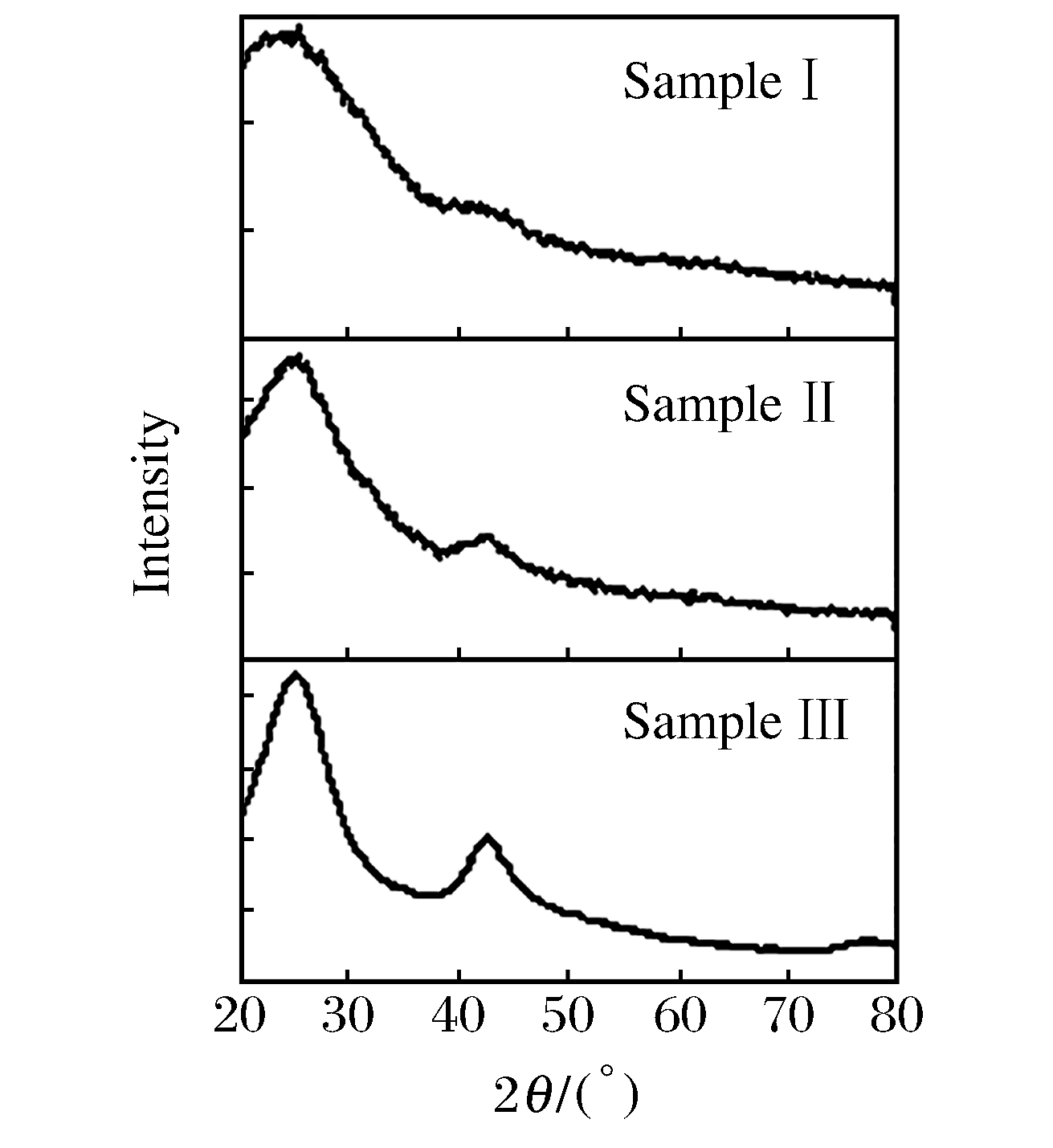
Sample mH3BO3:mC3N6H6 Full width at half maximum/(°) Product color xB:xC:xN Ⅰ 1:2 12.0 Black 28:50:22 Ⅱ 1:1 7.8 Tawny 41:25:34 Ⅲ 2:1 7.5 White 29:53:18 表 2 样品的实验条件和冲击压力
Table 2. Experimental conditions and shock pressure of samples
Sample Initial density/(g/cm3) Impact velocity/(km/s) Shock pressure/(GPa) Shock temperature/(K) Ⅰ 6.68 1.70 40 2 200 Ⅱ 6.59 1.70 40 2 300 Ⅲ 6.43 1.70 40 2 500 -
[1] Cheng Y C, Wu X L, Li S H, et al. Ab initio determination of lattice dynamics and thermodynamics of β-BC2N[J]. Solid State Commun, 2008, 146(1/2): 69-72. http://www.mendeley.com/research/ab-initio-determination-lattice-dynamics-thermodynamics-bbc2n/ [2] Langenhorst F, Solozhenko V L. ATEM-EELS study of new diamond-like phases in the B-C-N system[J]. Phys Chem Chem Phys, 2002, 4(20): 5183-5188. doi: 10.1039/B206691B [3] Wu B R, Huang Z Q, Su W S, et al. Atomic structure and mechanical properties of BC2N superlattice[J]. Diam Relat Mater, 2010, 19(11): 1341-1347. doi: 10.1016/j.diamond.2010.06.022 [4] Sun H, Jhi S H, Roundy D, et al. Structural forms of cubic BC2N[J]. Phys Rev B, 2001, 64(9): 094108. doi: 10.1103/PhysRevB.64.094108 [5] Zhang Y, Sun H, Chen C. Superhard cubic BC2N compared to diamond[J]. Phys Rev Lett, 2004, 93(19): 195504. doi: 10.1103/PhysRevLett.93.195504 [6] Luo X G, Guo X J, Xu B, et al. Body-centered superhard BC2N phases from first principles[J]. Phys Rev B, 2007, 76(9): 094103. doi: 10.1103/PhysRevB.76.094103 [7] Caretti I, Jimenez I, Albella J M. BCN films with controlled composition obtained by the interaction between molecular beams of B and C with nitrogen ion beams[J]. Diam Relat Mater, 2003, 12: 1079-1083. doi: 10.1016/S0925-9635(02)00276-5 [8] Mannan M A, Noguchi H, Kida T, et al. Growth and characterization of stoichiometric BCN films on highly oriented pyrolytic graphite by radiofrequency plasma enhanced chemical vapor deposition[J]. Thin Solid Films, 2010, 518(15): 4163-4169. doi: 10.1016/j.tsf.2009.11.086 [9] Nakano S, Akaishi M, Sasaki T, et al. Segregative crystallization of several diamond-like phases from the graphitic BC2N without an additive at 7.7 GPa[J]. Chemistry Mater, 1994, 6(12): 2246-2251. doi: 10.1021/cm00048a011 [10] Solozhenko V L, Andrault D, Fiquet G, et al. Synthesis of superhard cubic BC2N[J]. Appl Phys Lett, 2001, 78(10): 1385-1387. doi: 10.1063/1.1337623 [11] 何巨龙, 田永君, 于栋利, 等.高压合成B-C-N化合物的结构表征[J].硅酸盐学报, 2002, 30(1): 51-56. http://www.cqvip.com/Main/Detail.aspx?id=6018634He J L, Tian Y J, Yu D L, et al. Structure characterization of high-pressure-synthesized B-C-N compounds[J]. Journal of the Chinese Ceramic Society, 2002, 30(1): 51-56. (in Chinese) http://www.cqvip.com/Main/Detail.aspx?id=6018634 [12] 胡文涛.晶态BCN化合物的高温高压合成及其热膨胀性质[D].秦皇岛: 燕山大学, 2004.Hu W T. Synthesis of crystalline BCN compounds under high pressure and high temperature and their thermal expansion property[D]. Qinhuangdao: Yanshan University, 2004. (in Chinese) [13] 穆云超.硼碳氮晶体的制备工艺研究[D].郑州: 郑州大学, 2006.Mu Y C. Research for boron carbonitride crystals preparation[D]. Zhengzhou: Zhengzhou University, 2006. (in Chinese) [14] 白锁柱. BCN化合物的合成、表征和性能[D].长春: 吉林大学, 2005.Bai S Z. Synthesis, characterization and properties of boron carbon nitride compounds[D]. Changchun: Jilin University, 2005. (in Chinese) [15] 李雪飞, 张剑, 沈龙海, 等.六角相硼碳氮化合物的合成研究[J].高压物理学报, 2007, 21(3): 237-241. http://d.wanfangdata.com.cn/Periodical/gywlxb200703003Li X F, Zhang J, Shen L H, et al. Synthesis and characterization of h-BCN nanocrystallite under high-pressure and high-temperature[J]. Chinese Journal of High Pressure Physics, 2007, 21(3): 237-241. (in Chinese) http://d.wanfangdata.com.cn/Periodical/gywlxb200703003 [16] 杨建, 丘泰, 沈春英, 等.先驱体热解制备富硼、氮BCN化合物及其发光性能研究[J].无机材料学报, 2009, 24(1): 13-17. http://www.cqvip.com/QK/93432X/200901/29194693.htmlYang J, Qiu T, Shen C Y, et al. Photoluminescence of(boron and nitrogen)-rich BCN compounds pyrolysed from precursor[J]. Joumal of Inorganic Materials, 2009, 24(1): 13-17. (in Chinese) http://www.cqvip.com/QK/93432X/200901/29194693.html [17] Matizamhuka W R. A study on the synthesis of ultrahard cubic BC2N heterodiamond[D]. Johannesburg, South Africa: University of the Witwatersrand, 2010. [18] 马海云, 刘福生, 李永宏, 等.强冲击压缩条件下g-C3N4向β-C3N4直接转化[J].高压物理学报, 2012, 26(3): 319-324. http://www.cnki.com.cn/Article/CJFDTotal-GYWL201203014.htmMa H Y, Liu F S, Li Y H, et al. Strong shock-compression of g-C3N4 precursor for direct synthesis of β-C3N4[J]. Chinese Journal of High Pressure Physics, 2012, 26(3): 319-324. (in Chinese) http://www.cnki.com.cn/Article/CJFDTotal-GYWL201203014.htm [19] 白锁柱, 姚斌, 黄保坤, 等. BCN化合物的合成与表征[J].高等学校化学学报, 2005, 26(5): 811-815. http://www.cqvip.com/Main/Detail.aspx?id=20722950Bai S Z, Yao B, Huang B K, et al. Preparation and characterization of BCN[J]. Chemical Journal of Chinese Universities, 2005, 26(5): 811-815. (in Chinese) http://www.cqvip.com/Main/Detail.aspx?id=20722950 [20] 经福谦.实验物态方程导引[M].第2版.北京: 科学出版社, 1999: 199-200.Jing F Q. Introduction to Experimental Equation of State[M]. 2nd Ed. Beijing: Science Press, 1999: 199-200. (in Chinese) [21] Onodera A, Matsumoto K, Hirai T, et al. Synthesis of dense forms of B-C-N system using chemical-vapor-deposition/high-pressure process[J]. J Mater Sci, 2001, 36(3): 679-684. doi: 10.1023/A:1004832807084 [22] Watanabe M O, Itoh S, Mizushima K, et al. Bonding characterization of BC2N thin films[J]. Appl Phys Lett, 1996, 68(21): 2962-2964. doi: 10.1063/1.116369 -







 下载:
下载: