Properties of Surfactant-Modified Ammonium Nitrate
-
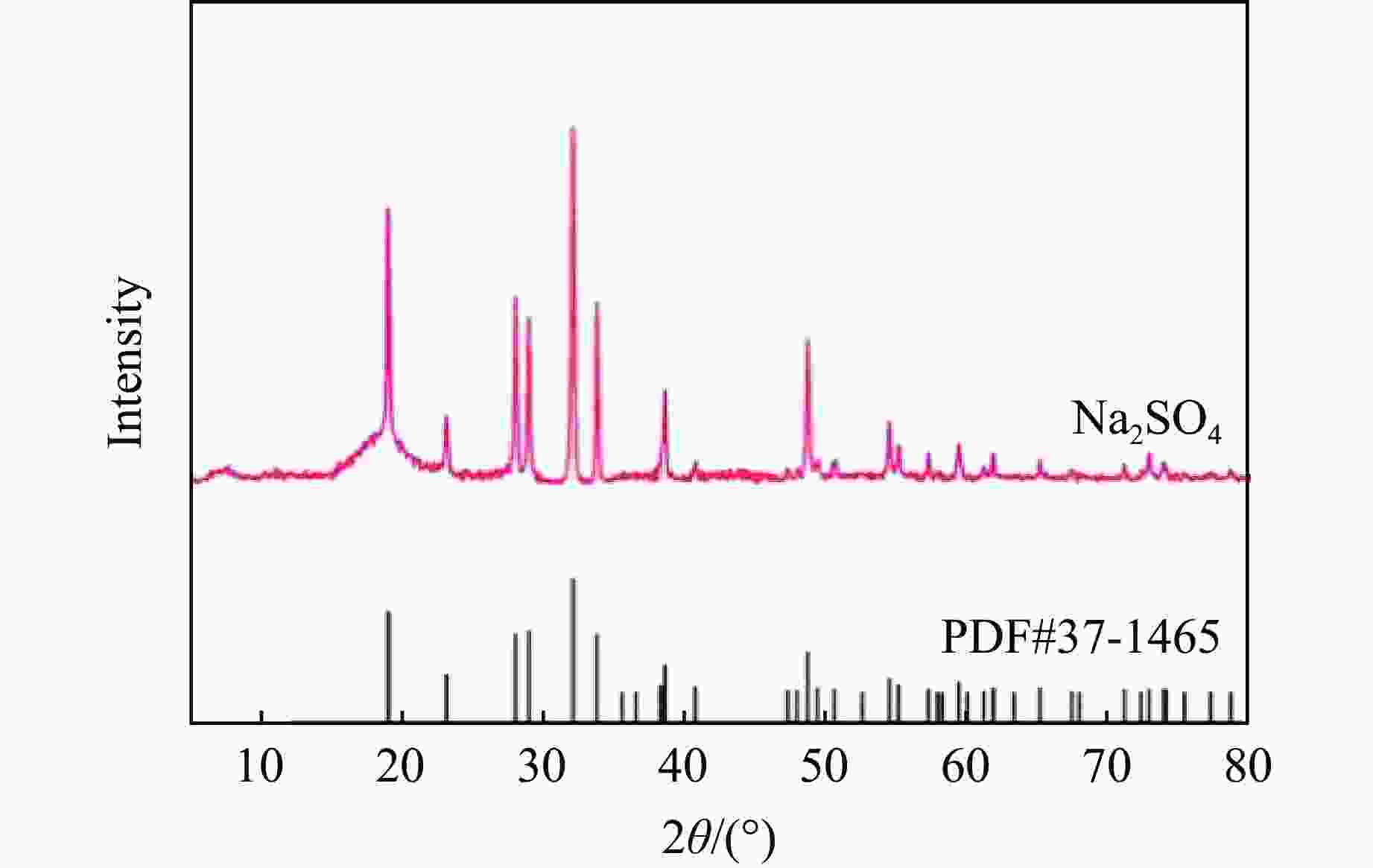
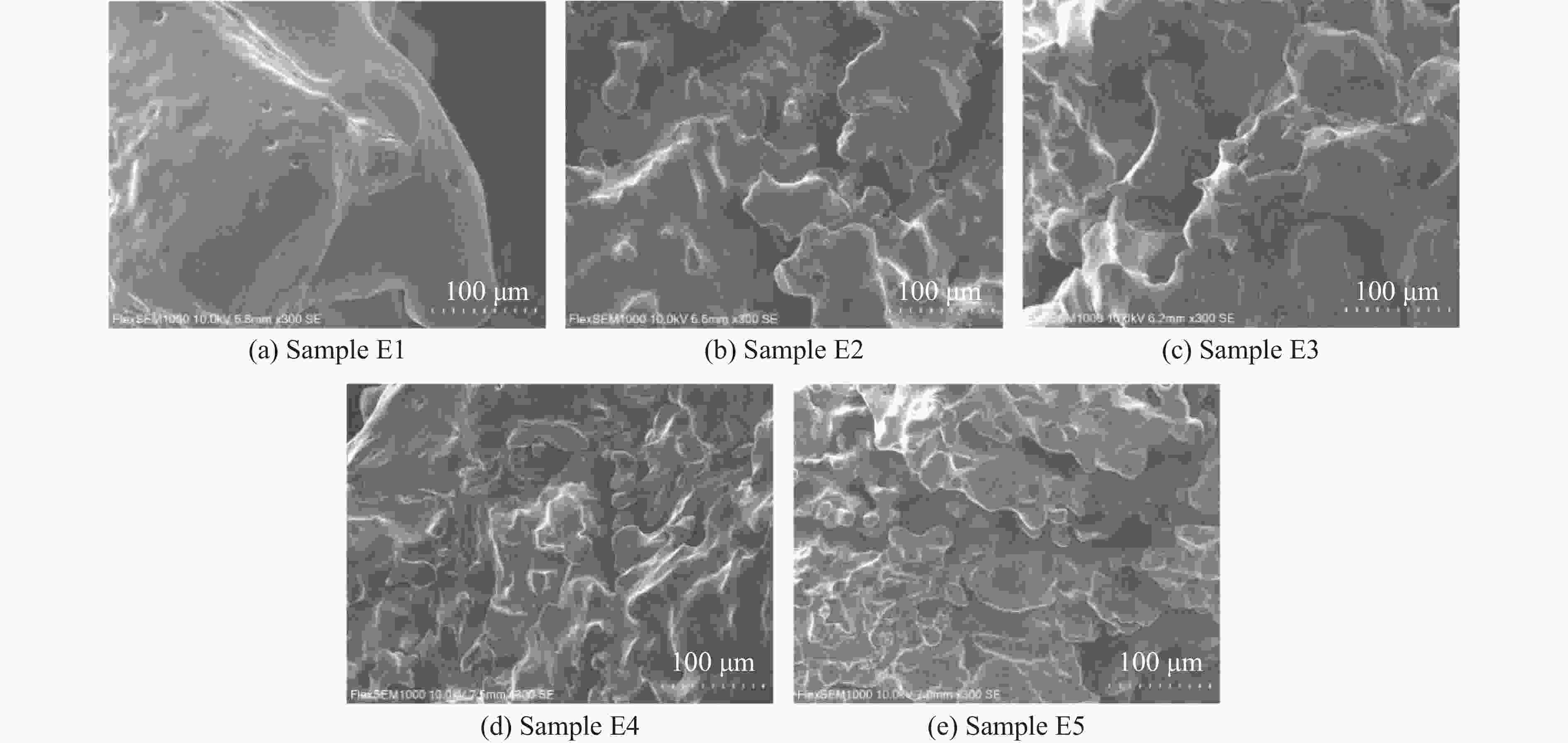
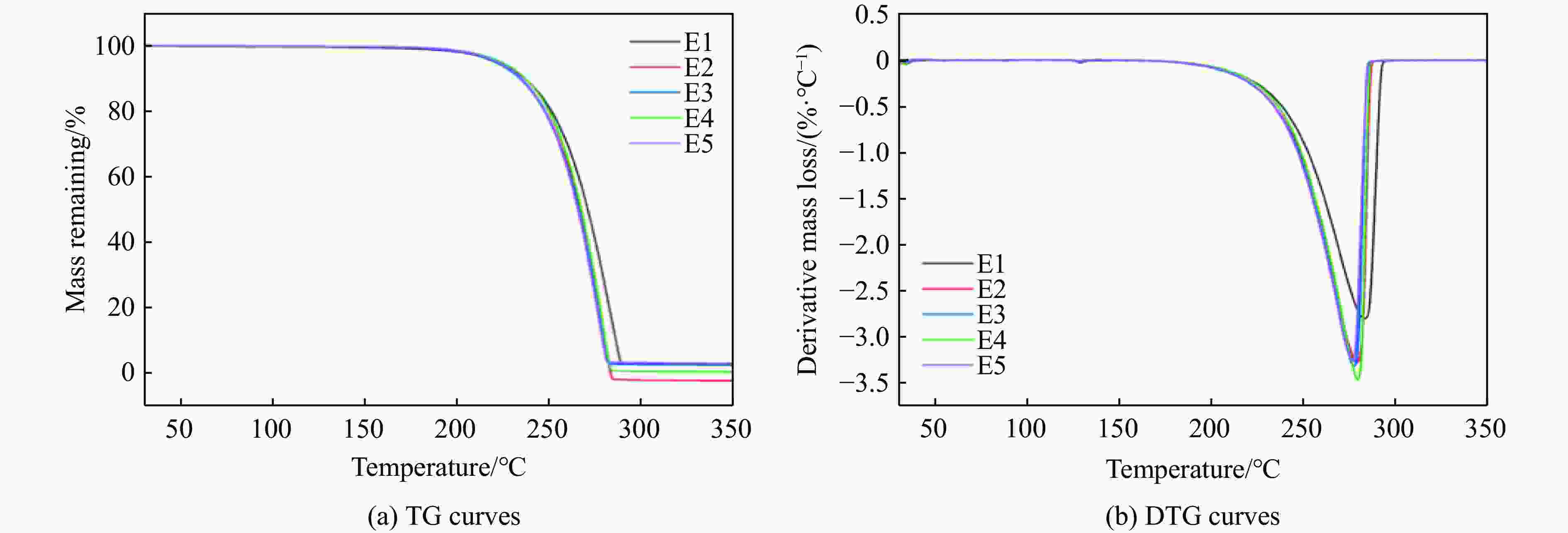
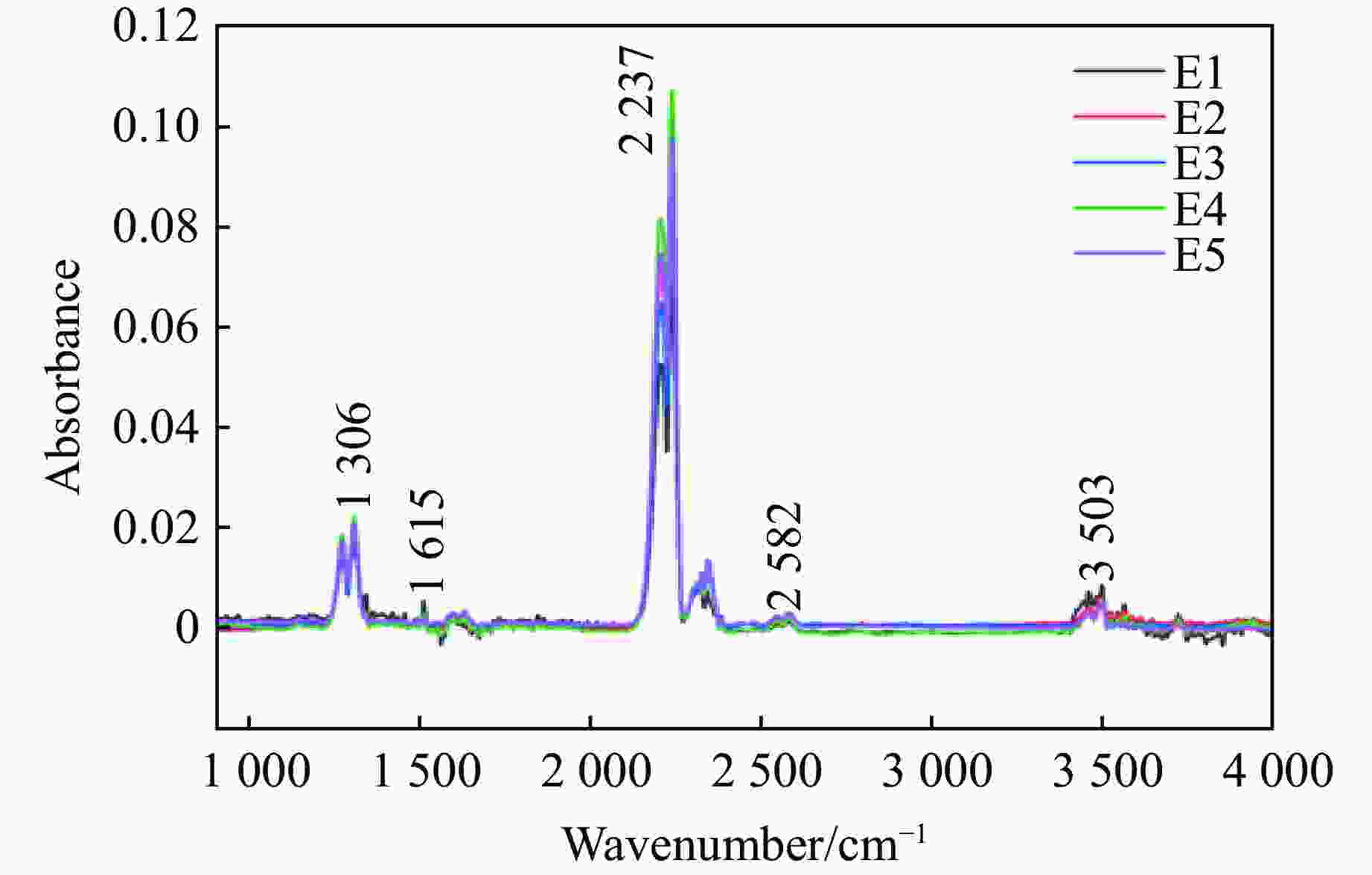
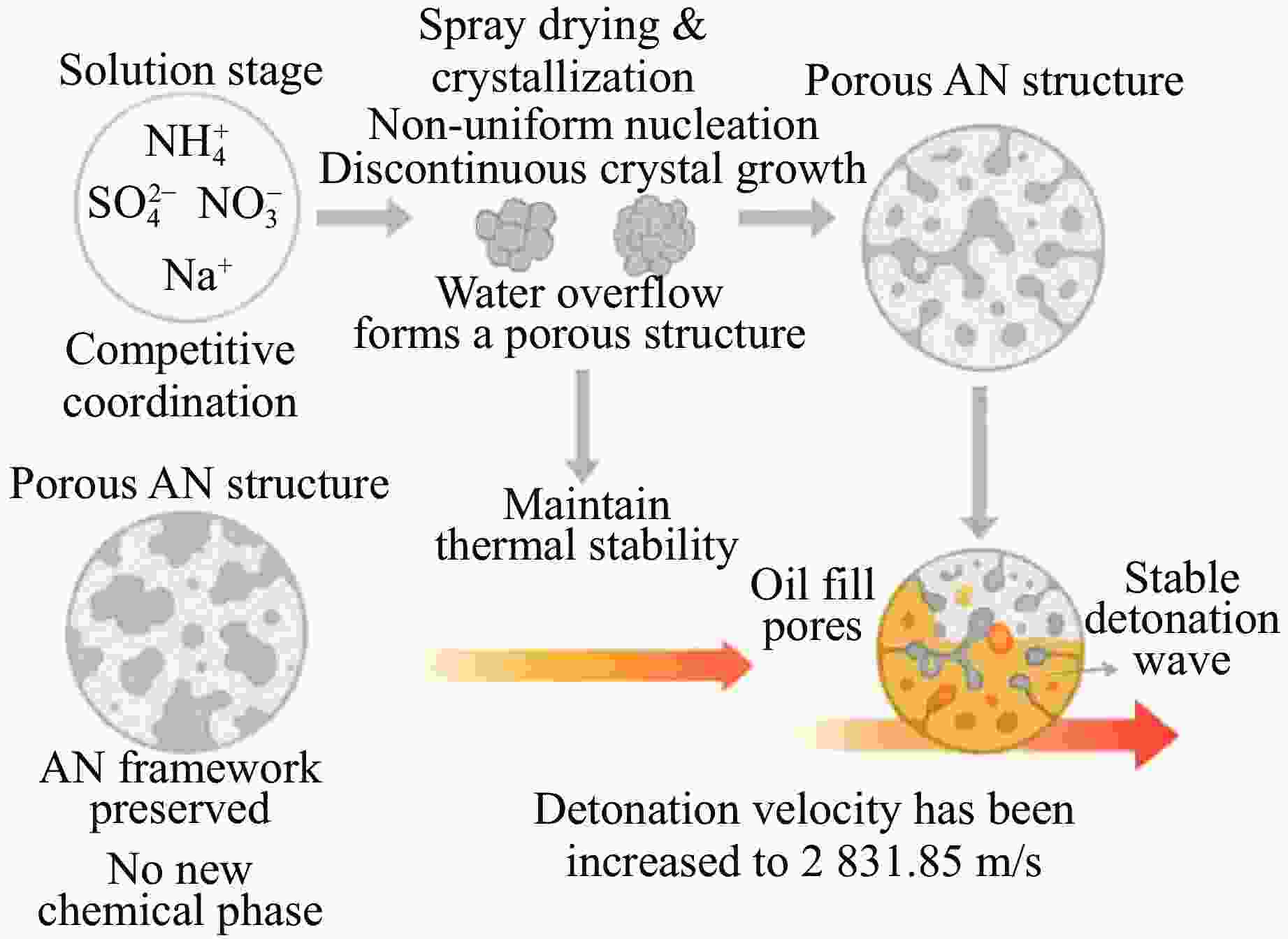
摘要: 多孔硝酸铵因其多孔隙结构常被用于特定作业,但其运输成本较高。为此,以离子表面活性剂PST为添加剂,通过喷雾造粒法制备了多孔隙改性硝酸铵,研究了不同质量分数(0~0.4%)的PST对硝酸铵孔隙结构、吸油率、热稳定性及爆炸性能的影响。结果表明:随着PST含量的增加,硝酸铵由致密颗粒逐步转变为具有明显连通孔隙的多孔结构,热稳定性基本保持不变,基体化学组成未发生本质改变,但吸附水降低。改性后样品与油相的结合能力提高,所配装药的爆速由未改性时的“未正常起爆”提升至
2831.85 m/s。微量PST可在基本不损害热安全性的前提下,诱导硝酸铵形成多孔结构,并显著改善爆速表现,具有较大的工程应用潜力。Abstract: Porous ammonium nitrate is frequently used for specific applications due to its porous structure when compared with conventional ammonium nitrate, however, its higher transportation costs increase overall operational expenses. This study investigated the preparation of porous granular modified ammonium nitrate using ionic surfactant PST as an additive via spray granulation. The effects of varying PST concentrations (0−0.4%) on the pore structure, oil absorption capacity, thermal stability, and explosive properties of ammonium nitrate were examined. The research results indicate that increasing PST content gradually transforms dense ammonium nitrate particles into a porous structure with distinct interconnected pores. Thermal stability remains essentially unchanged, and the matrix chemical composition undergoes no fundamental alteration, though its adsorbed water content decreases. The modified samples exhibit enhanced binding capacity with the oil phase. The detonation velocity of the assembled charge increases from “failed to detonate normally” in the unmodified state to2831.85 m/s. Trace amounts of PST can induce the formation of a porous structure in ammonium nitrate without significantly compromising thermal safety, while markedly improving detonation velocity performance, demonstrating potential for engineering applications.-
Key words:
- modified ammonium nitrate /
- spray granulation /
- porosity /
- thermal stability /
- explosive properties
-
表 1 改性硝酸铵溶液配方
Table 1. Modified ammonium nitrate solution formulation
No. Mass fraction of PST/% Mass fraction of AN/% Mass fraction of H2O/% E1 0 60.00 40 E2 0.06 59.94 40 E3 0.12 59.88 40 E4 0.18 59.82 40 E5 0.24 59.76 40 -
[1] MIYAKE A, TAKAHARA K, OGAWA T, et al. Influence of physical properties of ammonium nitrate on the detonation behaviour of ANFO [J]. Journal of Loss Prevention in the Process Industries, 2001, 14(6): 533–538. doi: 10.1016/S0950-4230(01)00041-9 [2] 刘松华. 工业硝酸铵的改性研究 [D]. 南京: 南京理工大学, 2012.LIU S H. The modification studies of industrial ammonium nitrate [D]. Nanjing: Nanjing University of Science & Technology, 2012. [3] OXLEY J C, SMITH J L, ROGERS E, et al. Ammonium nitrate: thermal stability and explosivity modifiers [J]. Thermochimica Acta, 2002, 384: (1/2): 23–45. [4] 陈阳, 康自华, 陈婷, 等. 尿素和硫酸铵对多孔硝酸铵结构改性的影响研究 [J]. 火工品, 2024(6): 40–47. doi: 10.3969/j.issn.1003-1480.2024.06.008CHEN Y, KANG Z H, CHEN T, et al. Study on the effects of urea and ammonium sulfate on the structural modification of porous ammonium nitrate [J]. Initiators & Pyrotechnics, 2024(6): 40–47. doi: 10.3969/j.issn.1003-1480.2024.06.008 [5] 刘博. 溶剂挥发法改性硝酸铵防吸湿性能研究 [D]. 兰州: 兰州理工大学, 2024.LIU B. Anti-hygroscopic properties of ammonium nitrate modified by solvent evaporation method [D]. Lanzhou: Lanzhou University of Science and Technology, 2024. [6] 张洪. 改性硝酸铵在现场混装中的应用研究 [D]. 淮南: 安徽理工大学, 2020.ZHANG H. Application of modified ammonium nitrate in field mixed loading [D]. Huainan: Anhui University of Science and Technology, 2020. [7] 郭紫卿, 唐秋明. 多孔粒状硝酸铵的表面膨化技术及应用 [J]. 爆破器材, 2025, 54(3): 27–33. doi: 10.3969/j.issn.1001-8352.2025.03.005GUO Z Q, TANG Q M. Surface expansion technology and the application of porous granular ammonium nitrate [J]. Explosive Materials, 2025, 54(3): 27–33. doi: 10.3969/j.issn.1001-8352.2025.03.005 [8] 戴礼, 张洪秀, 周莹, 等. 新型表面活性剂改性硝酸铵的研究 [J]. 化学工程与装备, 2017(11): 41–42. doi: 10.19566/j.cnki.cn35-1285/tq.2017.11.012DAI L, ZHANG H X, ZHOU Y, et al. Research on modification of ammonium nitrate using novel surfactants [J]. Chemical Engineering & Equipment, 2017(11): 41–42. doi: 10.19566/j.cnki.cn35-1285/tq.2017.11.012 [9] 叶志文. Gemini表面活性剂C12-3(OH)-C12·2Cl对膨化硝酸铵晶体性能影响的研究 [J]. 精细与专用化学品, 2006, 14(Suppl 1): 42–45. doi: 10.3969/j.issn.1008-1100.2006.z1.012YE Z W. Research on influence of Gemini surfactant C12-3(OH)-C12·2Cl on crystal properties of expanded ammonium nitrate [J]. Fine and Specialty Chemcals, 2006, 14(Suppl 1): 42–45. doi: 10.3969/j.issn.1008-1100.2006.z1.012 [10] BORIS G, ABRAHAMS, EVGENYZ, et al. Method for preparing an encapsulated slow-release particulate fertilizer: US5560768 [P]. 1996-10-01. [11] ZYGMUNT B, BUCZKOWSKI D. Influence of ammonium nitrate prills’ properties on detonation velocity of ANFO [J]. Propellants, Explosives, Pyrotechnics, 2007, 32(5): 411–414. doi: 10.1002/prep.200700045 [12] 尚德忠, 耿立君, 马宏伟. 铵油炸药在露天矿煤层爆破中的应用 [J]. 露天采矿技术, 2003(1): 16, 26.SHANG D Z, GENG L J, MA H W. Application of ammonium nitrate fuel oil mixture to coal seam blasting of open-pit coal mine [J]. Opencast Mining Technology, 2003(1): 16, 26. [13] 宋占辉. 浅析ANFO铵油炸药在隧道工程中的应用 [J]. 中国新技术新产品, 2015(3): 186–187. doi: 10.13612/j.cnki.cntp.2015.03.162SONG Z H. A brief analysis of the application of ANFO ammonium oil explosives in tunnel engineering [J]. China New Technologies and Products, 2015(3): 186–187. doi: 10.13612/j.cnki.cntp.2015.03.162 [14] 李云岗. 利用多孔粒状硝酸铵装置生产高密度硝酸铵产品 [J]. 大氮肥, 2020, 43(6): 402–404. doi: 10.3969/j.issn.1002-5782.2020.06.011LI Y G. Use porous granular ammonium nitrate plant to produce high density ammonium nitrate [J]. Large Scale Nitrogenous Fertilizer Industry, 2020, 43(6): 402–404. doi: 10.3969/j.issn.1002-5782.2020.06.011 [15] 聂凡, 仝坤, 邵志国, 等. 重质有机质热解过程中挥发物的产出与逸出气体分析研究进展 [J]. 化工进展, 2022, 41(5): 2322–2331. doi: 10.16085/j.issn.1000-6613.2021-1009NIE F, TONG K, SHAO Z G, et al. Volatile generation during pyrolysis of heavy organic matters and the development of the evolution gas analysis [J]. Chemical Industry and Engineering Progress, 2022, 41(5): 2322–2331. doi: 10.16085/j.issn.1000-6613.2021-1009 [16] 杨文军. 关于多孔粒状铵油炸药性能的研究 [J]. 山西化工, 2023, 43(9): 31–32. doi: 10.16525/j.cnki.cn14-1109/tq.2023.09.013YANG W J. Research on porous granular ammonium oil explosives in the chemical industry [J]. Shanxi Chemical Industry, 2023, 43(9): 31–32. doi: 10.16525/j.cnki.cn14-1109/tq.2023.09.013 [17] 林木良. 热重-红外联用方法在药物热降解和热氧降解研究上的应用 [J]. 广州化工, 2002, 30(3): 44–47. doi: 10.3969/j.issn.1001-9677.2002.03.014LIN M L. Applications of TG-IR on thermal degradation and thermal oxidation degradation of medicine [J]. Guangzhou Chemical Industry, 2002, 30(3): 44–47. doi: 10.3969/j.issn.1001-9677.2002.03.014 [18] DUNUWILLE M, YOO C S. Phase diagram of ammonium nitrate [J]. The Journal of Chemical Physics, 2013, 139(21): 214503. doi: 10.1063/1.4837715 -






 下载:
下载: