Metallic Hydrogen Ligand Compounds: A Potential Route to Superconducting Metallic Hydrogen at Ambient Pressure
-
摘要: 金属氢因具有室温超导电性、量子流体等特性,被认为是高压物理研究领域的“圣杯”。理论研究认为,获得原子化的金属氢需要500 GPa以上压强,因此,自从1935年金属氢概念被提出以来,至今尚未在实验中获得。为使人们能够在生产和生活中利用金属氢的特殊性质,需要在环境压力下获得金属氢。目前,低压下获得金属氢的研究思路是利用氢化物的“化学预压缩”让氢在较低压力下进行金属化,即寻找特殊的氢化物作为金属氢性质的宿主。但是,这类材料至今没有明显的结构特征,不仅加大了探索金属氢宿主材料的难度,还打击了人们在环压力下获得金属氢性质的信心。为此,科研人员又提出了以氢为配体的高配位氢化物—低压下金属氢性质的潜在宿主,其中,配体氢的非键轨道是否金属化是氢配体化合物能否承载金属氢性质的判断标准。首先,总结了氢在常压下的主要行为,重点关注常压下的氢配体化合物;然后,通过一维氢原子链这一简单模型,分析非键轨道金属化的原因和降低稳定压力的物理图像;接着,分析金属氢配体化合物的轨道特点,指出其超导电性、拓扑性质和实现金属化的电子结构规律。所提出的关于金属氢配体化合物的分析,不仅为未来探索金属氢化物超导体提供了重要的结构信息,更为人们在环境压力下获得金属氢的性质提供了重要的理论基础。Abstract: Metallic hydrogen, with its properties including room-temperature superconductivity and quantum fluidity, is known as the holy grail of high-pressure physics. However, since atomic metallic hydrogen requires pressures about 500 GPa, it has not been realized in experiments since its conception in 1935. To take advantage of properties the properties of metallic hydrogen in the future, it will be crucial to obtain it at ambient pressure. Current approaches to obtaining metallic hydrogen at low pressures rely on the “chemical precompression” in hydrides to induce metallization of hydrogen at low pressures, essentially identifying superconducting hydrides that can host the properties of metallic hydrogen. However, these superconducting hydrides currently lack distinct structural features, complicating the search for metallic hydrogen hosts. Here, we identify metallic hydrogen ligand compounds with hydrogen as the ligands as potential hosts for properties of metallic hydrogen at low pressures. The metallization of the non-bonding orbitals of the hydrogen ligands is a key criterion for determining whether a metallic hydrogen ligand compound can host metallic hydrogen properties. This article summarizes the main behaviors of hydrogen at ambient pressure, focusing on hydrogen ligand compounds at ambient pressure. Then, using a simple model of a one-dimensional hydrogen atom chain, we analyzed the causes of non-bonding orbital metallization and the physical picture of reduced stability pressure. The orbital characteristics of metallic hydrogen ligand compounds are then analyzed, highlighting their rules of superconductivity, topological properties, and the electronic structure that enable metallization. The analysis of metallic hydrogen ligand compounds presented in this article not only provides important structural information for future exploration of metal hydride superconductors but also provides an important theoretical foundation for realizing the properties of metallic hydrogen at ambient pressure.
-
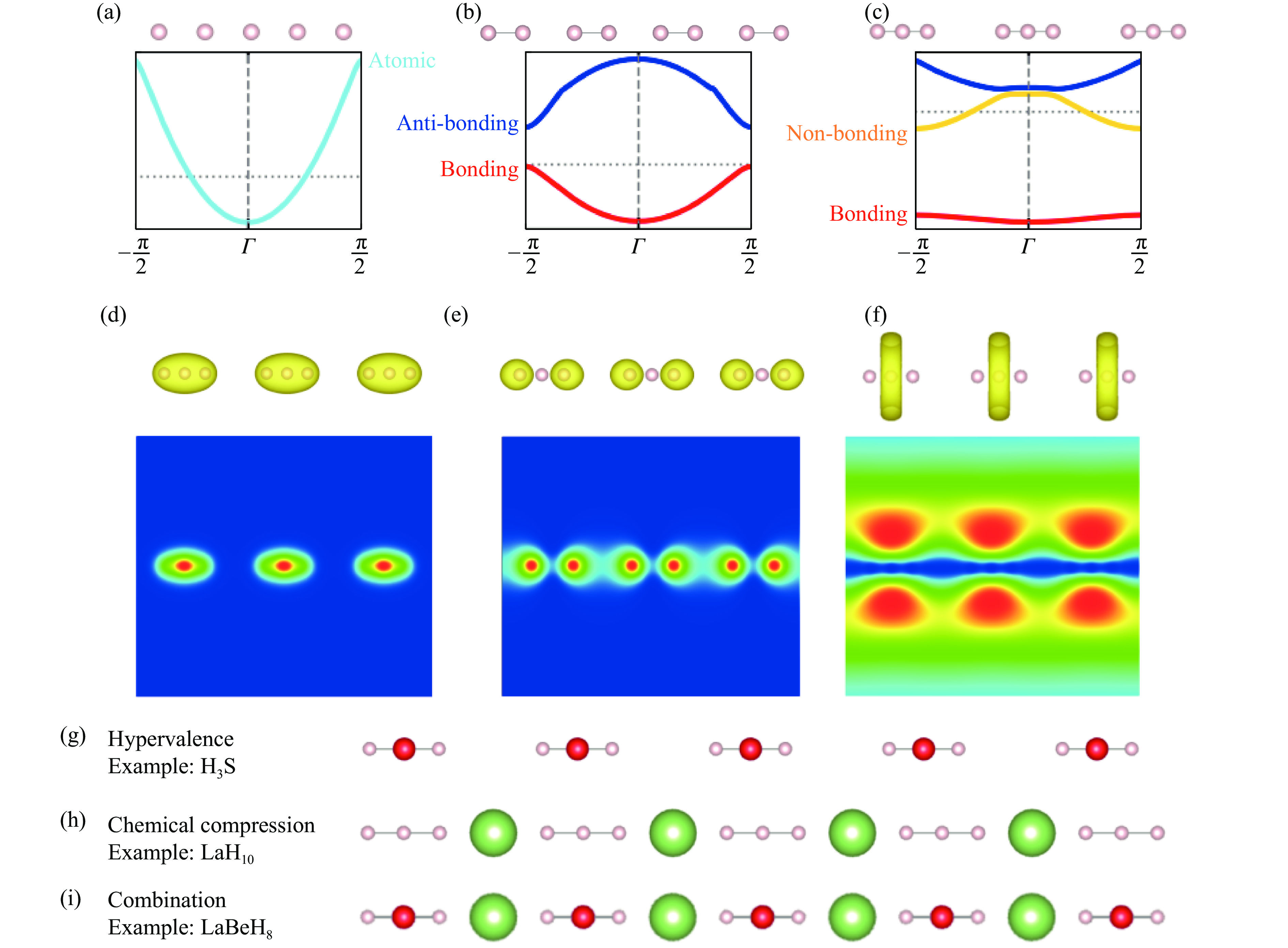
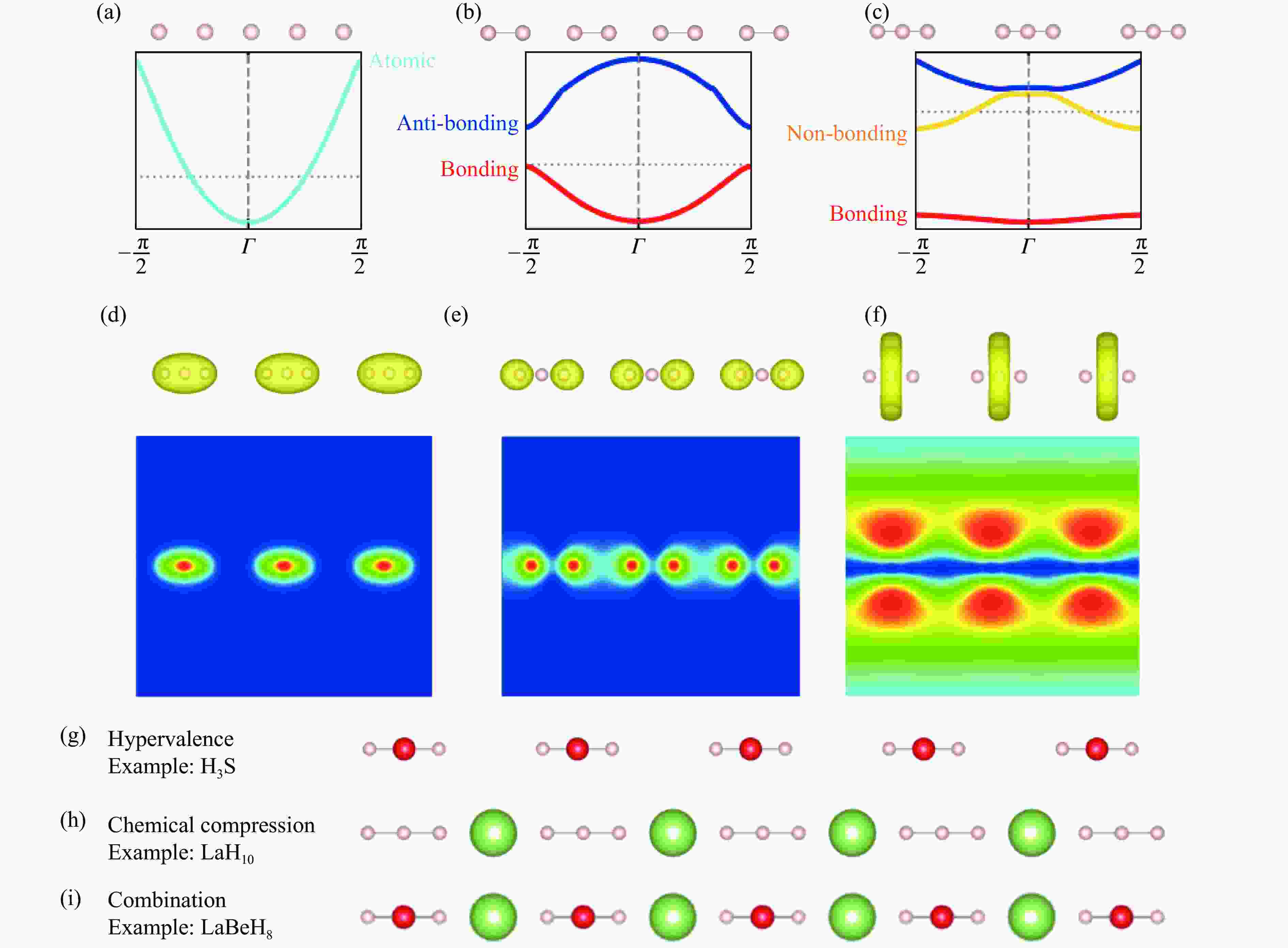
图 1 原子化、二聚化和三聚化的一维无限氢原子链模型:(a) 原子化、(b) 二聚化和(c) 三聚化的一维氢原子链晶体结构和电子结构,三聚化一维氢原子链的(d) 成键轨道、(e) 非键轨道和(f) 反键轨道的电子密度,降低氢金属化稳定压力途径在一维原子链中的 (g) 三中心一维超价氢化物链、(h) 阳离子化学压缩的一维三聚化氢原子基团、(i) 阳离子化学压缩的三中心一维超价氢原子链
Figure 1. Atomized, dimerized, and trimerized one-dimensional infinite hydrogen-atom-chain models: crystal structure and electronic structure of (a) atomized, (b) dimerized, and (c) trimerized one-dimensional hydrogen atom chains; electron density of (d) bonding orbitals, (e) non-bonding orbitals, and (f) anti-bonding orbitals of trimerized one-dimensional hydrogen atom chain; the pathways for reducing the stability pressure of metallic hydrogen of one-dimensional atomic chains in (g) three-center one-dimensional hypervalent hydride chain, (h) one-dimensional trimerized hydrogen atom group compressed by cations, and (i) three-center one-dimensional hypervalent hydride chain compressed by cations
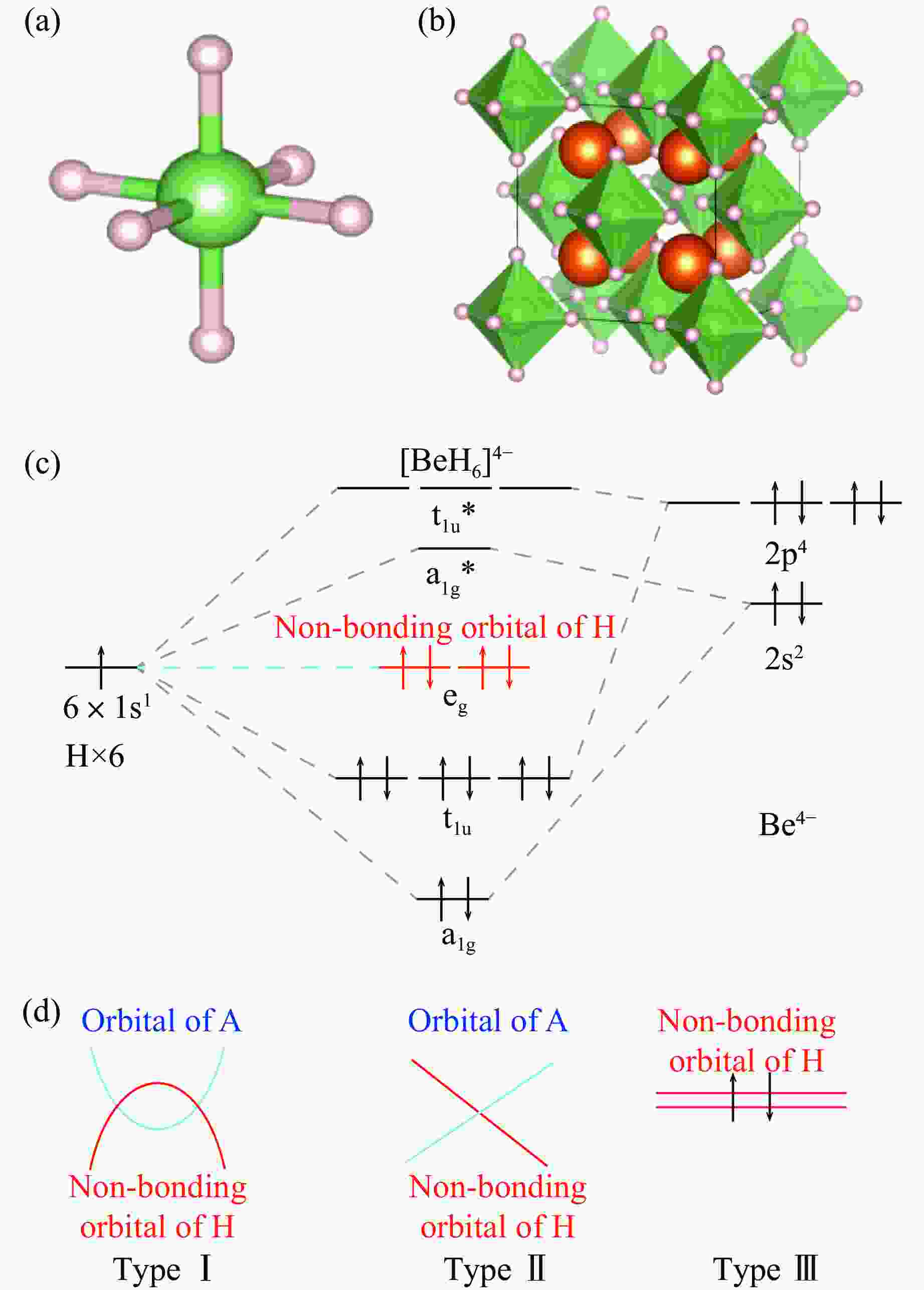
图 2 氢配体化合物Mg2BeH6的晶体和轨道以及潜在的拓扑电子结构[28–29]:(a) BeH6基团结构,(b) Mg2BeH6氢化物结构,(c) BeH6基团的轨道分析,(d) 与Mg2BeH6同构的氢化物潜在的电子拓扑
Figure 2. Crystals and orbitals of the hydrogen ligand compound Mg2BeH6 and potential topological electronic structures[28–29]: (a) structure of BeH6 group; (b) structure of Mg2BeH6 hydride; (c) orbital analysis of the BeH6 group; (d) potential electronic topology of the hydride isostructural to Mg2BeH6
-
[1] LUBITZ W, TUMAS W. Hydrogen: an overview [J]. Chemical Reviews, 2007, 107(10): 3900–3903. doi: 10.1021/cr050200z [2] MCMAHON J M, MORALES M A, PIERLEONI C, et al. The properties of hydrogen and helium under extreme conditions [J]. Reviews of Modern Physics, 2012, 84(4): 1607–1653. doi: 10.1103/RevModPhys.84.1607 [3] MONACELLI L, CASULA M, NAKANO K, et al. Quantum phase diagram of high-pressure hydrogen [J]. Nature Physics, 2023, 19(6): 845–850. doi: 10.1038/s41567-023-01960-5 [4] ASHCROFT N W. Metallic hydrogen: a high-temperature superconductor? [J]. Physical Review Letters, 1968, 21(26): 1748–1749. doi: 10.1103/PhysRevLett.21.1748 [5] ASHCROFT N W. Hydrogen dominant metallic alloys: high temperature superconductors? [J]. Physical Review Letters, 2004, 92(18): 187002. doi: 10.1103/PhysRevLett.92.187002 [6] WIGNER E, HUNTINGTON H B. On the possibility of a metallic modification of hydrogen [J]. The Journal of Chemical Physics, 1935, 3(12): 764–770. doi: 10.1063/1.1749590 [7] LI Y W, HAO J, LIU H Y, et al. The metallization and superconductivity of dense hydrogen sulfide [J]. The Journal of Chemical Physics, 2014, 140(17): 174712. doi: 10.1063/1.4874158 [8] BOGDANOVIĆ B, RITTER A, SPLIETHOFF B. Active MgH2-Mg systems for reversible chemical energy storage [J]. Angewandte Chemie International Edition, 1990, 29(3): 223–234. doi: 10.1002/anie.199002233 [9] OOYA K, LI J, FUKUI K, et al. Ruthenium catalysts promoted by lanthanide oxyhydrides with high hydride-ion mobility for low-temperature ammonia synthesis [J]. Advanced Energy Materials, 2021, 11(4): 2003723. doi: 10.1002/aenm.202003723 [10] MATSUOKA T, HISHIDA M, KUNO K, et al. Superconductivity of platinum hydride [J]. Physical Review B, 2019, 99(14): 144511. doi: 10.1103/PhysRevB.99.144511 [11] ERREA I, CALANDRA M, MAURI F. First-principles theory of anharmonicity and the inverse isotope effect in superconducting palladium-hydride compounds [J]. Physical Review Letters, 2013, 111(17): 177002. doi: 10.1103/PhysRevLett.111.177002 [12] EREMETS M I, TROJAN I A, MEDVEDEV S A, et al. Superconductivity in hydrogen dominant materials: silane [J]. Science, 2008, 319(5869): 1506–1509. doi: 10.1126/science.1153282 [13] MIAO M S, SUN Y H, ZUREK E, et al. Chemistry under high pressure [J]. Nature Reviews Chemistry, 2020, 4(10): 508–527. doi: 10.1038/s41570-020-0213-0 [14] DUAN D F, LIU Y X, TIAN F B, et al. Pressure-induced metallization of dense (H2S)2H2 with high-Tc superconductivity [J]. Scientific Reports, 2014, 4: 6968. doi: 10.1038/srep06968 [15] DROZDOV A P, EREMETS M I, TROYAN I A, et al. Conventional superconductivity at 203 kelvin at high pressures in the sulfur hydride system [J]. Nature, 2015, 525(7567): 73–76. doi: 10.1038/nature14964 [16] DROZDOV A P, KONG P P, MINKOV V S, et al. Superconductivity at 250 K in lanthanum hydride under high pressures [J]. Nature, 2019, 569(7757): 528–531. doi: 10.1038/s41586-019-1201-8 [17] LIU H Y, NAUMOV I I, HOFFMANN R, et al. Potential high-Tc superconducting lanthanum and yttrium hydrides at high pressure [J]. Proceedings of the National Academy of Sciences of the United States of America, 2017, 114(27): 6990–6995. doi: 10.1073/pnas.1704505114 [18] PENG F, SUN Y, PICKARD C J, et al. Hydrogen clathrate structures in rare earth hydrides at high pressures: possible route to room-temperature superconductivity [J]. Physical Review Letters, 2017, 119(10): 107001. doi: 10.1103/PhysRevLett.119.107001 [19] ZHAO W D, HUANG X L, ZHANG Z H, et al. Superconducting ternary hydrides: progress and challenges [J]. National Science Review, 2024, 11(7): nwad307. doi: 10.1093/nsr/nwad307 [20] ZHANG Z H, CUI T, HUTCHEON M J, et al. Design principles for high-temperature superconductors with a hydrogen-based alloy backbone at moderate pressure [J]. Physical Review Letters, 2022, 128(4): 047001. doi: 10.1103/PhysRevLett.128.047001 [21] SONG Y G, BI J K, NAKAMOTO Y, et al. Stoichiometric ternary superhydride LaBeH8 as a new template for high-temperature superconductivity at 110 K under 80 GPa [J]. Physical Review Letters, 2023, 130(26): 266001. doi: 10.1103/PhysRevLett.130.266001 [22] GAO K, CERQUEIRA T F T, SANNA A, et al. The maximum Tc of conventional superconductors at ambient pressure [J]. Nature Communications, 2025, 16(1): 8253. doi: 10.1038/s41467-025-63702-w [23] BARAN A, JENSEN T R, POLAŃSKI M. Synthesis of Mg2FeH6 by high-temperature high-pressure reactive planetary ball milling [J]. Advanced Engineering Materials, 2025, 27(6): 2400425. doi: 10.1002/adem.202400425 [24] PUHAKAINEN K, BENSON D, NYLÉN J, et al. Hypervalent octahedral ${{\mathrm{SiH_6^{2-} }}}$ species from high-pressure synthesis [J]. Angewandte Chemie International Edition, 2012, 51(13): 3156–3160. doi: 10.1002/anie.201108713 [25] BEARPARK M J, MCGRADY G S, PRINCE P D, et al. The first structurally characterized hypervalent silicon hydride: unexpected molecular geometry and Si−H···K interactions [J]. Journal of the American Chemical Society, 2001, 123(31): 7736–7737. doi: 10.1021/ja0162616 [26] XIE H, YAO Y S, FENG X L, et al. Hydrogen pentagraphenelike structure stabilized by hafnium: a high-temperature conventional superconductor [J]. Physical Review Letters, 2020, 125(21): 217001. doi: 10.1103/PhysRevLett.125.217001 [27] LIU P Y, ZHUANG Q, XU Q, et al. Mechanism of high-temperature superconductivity in compressed H2-molecular-type hydride [J]. Science Advances, 2025, 11(13): eadt9411. doi: 10.1126/sciadv.adt9411 [28] DOLUI K, CONWAY L J, HEIL C, et al. Feasible route to high-temperature ambient-pressure hydride superconductivity [J]. Physical Review Letters, 2024, 132(16): 166001. doi: 10.1103/PhysRevLett.132.166001 [29] ZHANG Z H, ZHU M F, SONG H, et al. Theoretical design of ellipsoidal nodal surface semimetals via hypervalent hydrides at high pressure [J]. Physical Review B, 2023, 108(20): 205119. doi: 10.1103/PhysRevB.108.205119 -






 下载:
下载: