Inhibition Mechanism of KHCO3-Containing Water Mist on Methane-Hydrogen Premixed Deflagration
-
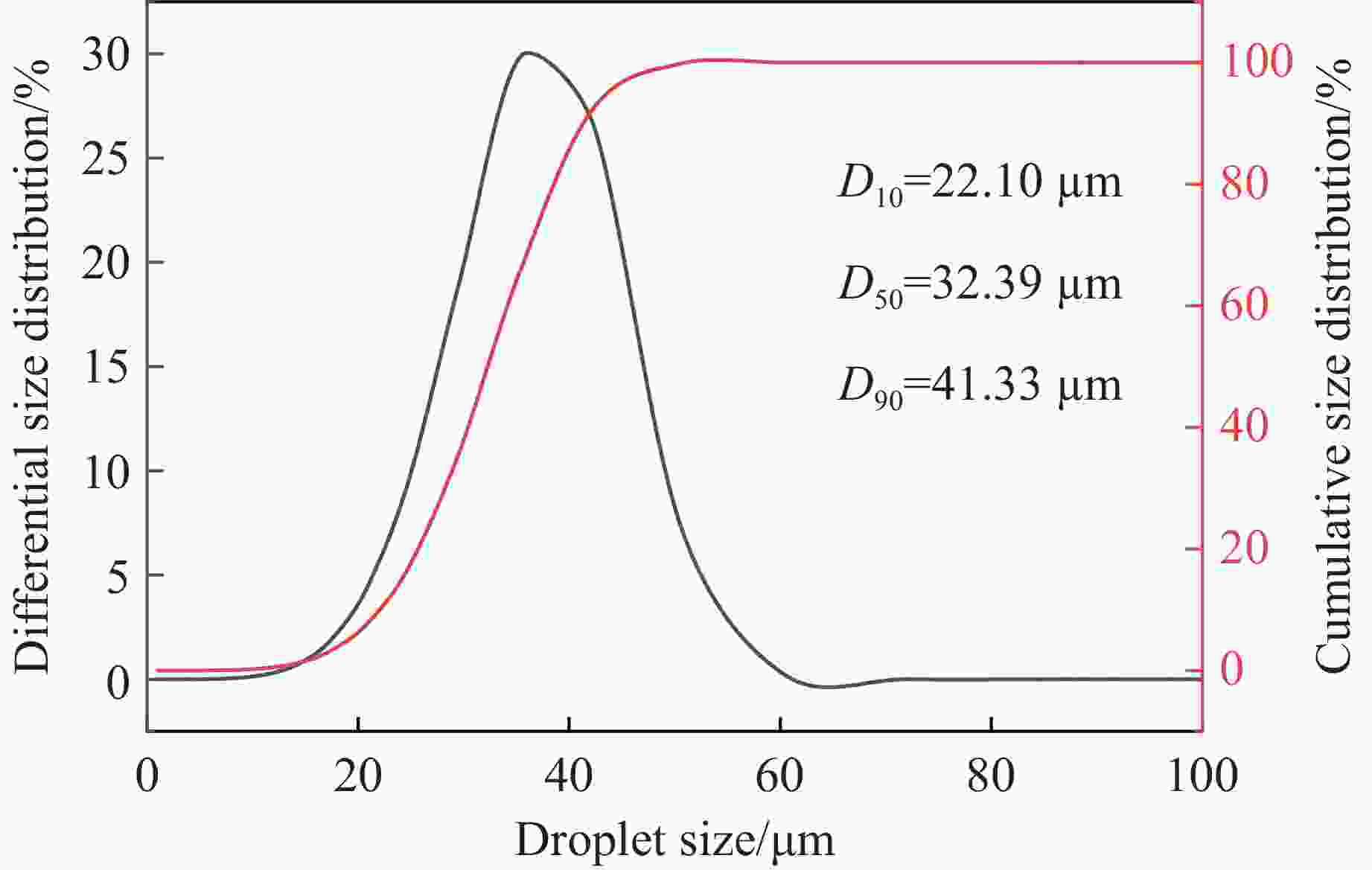
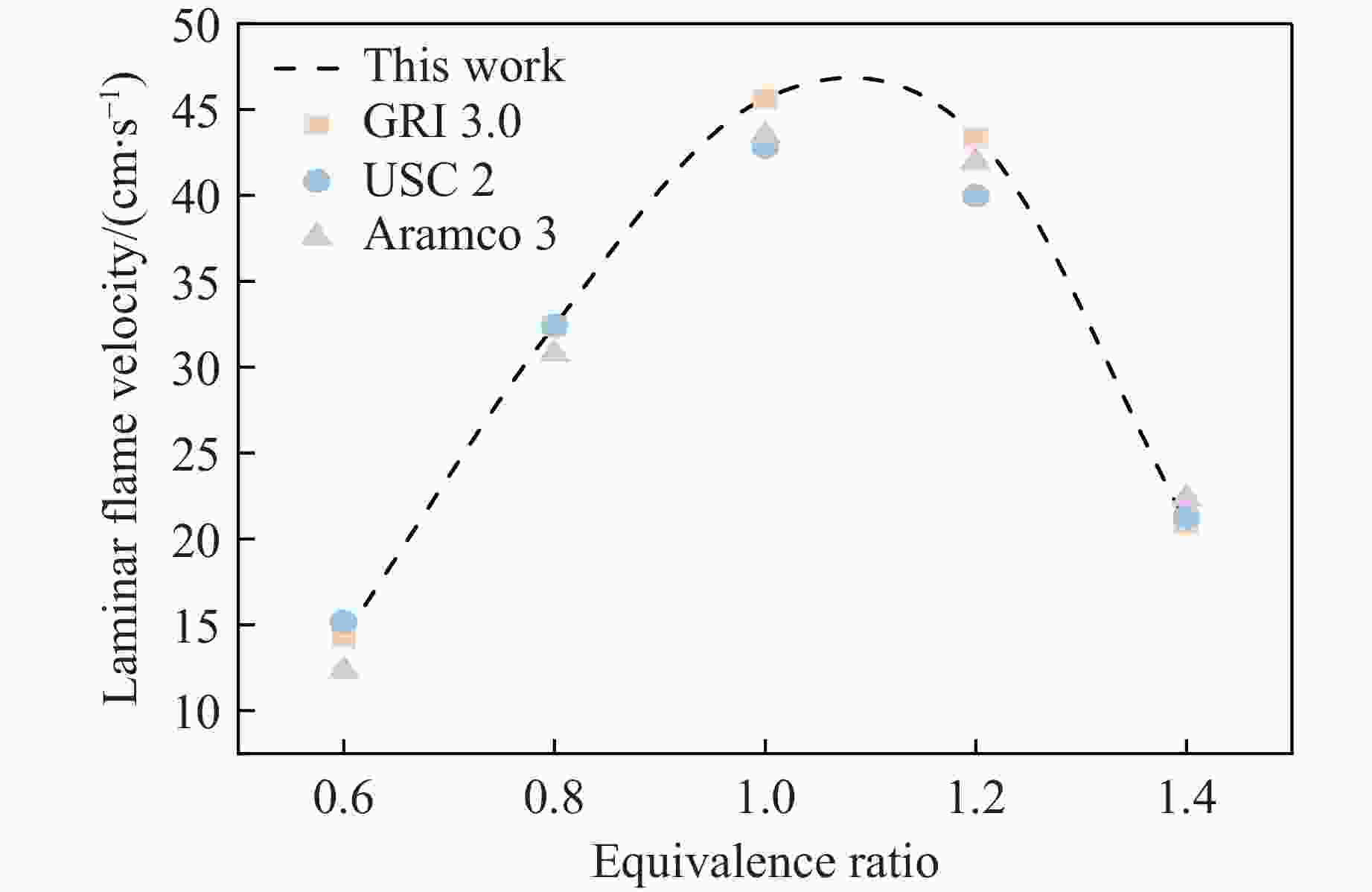
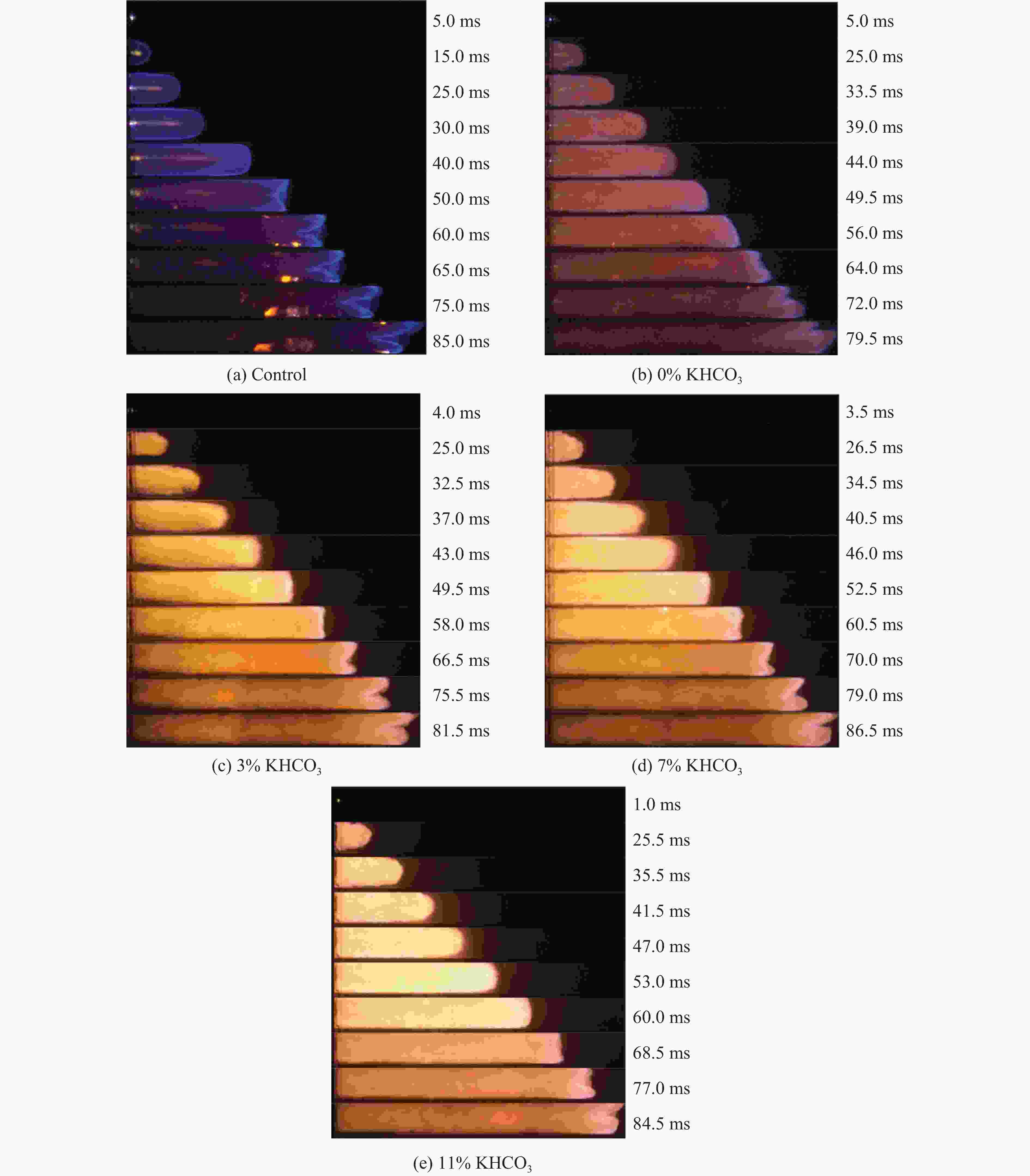
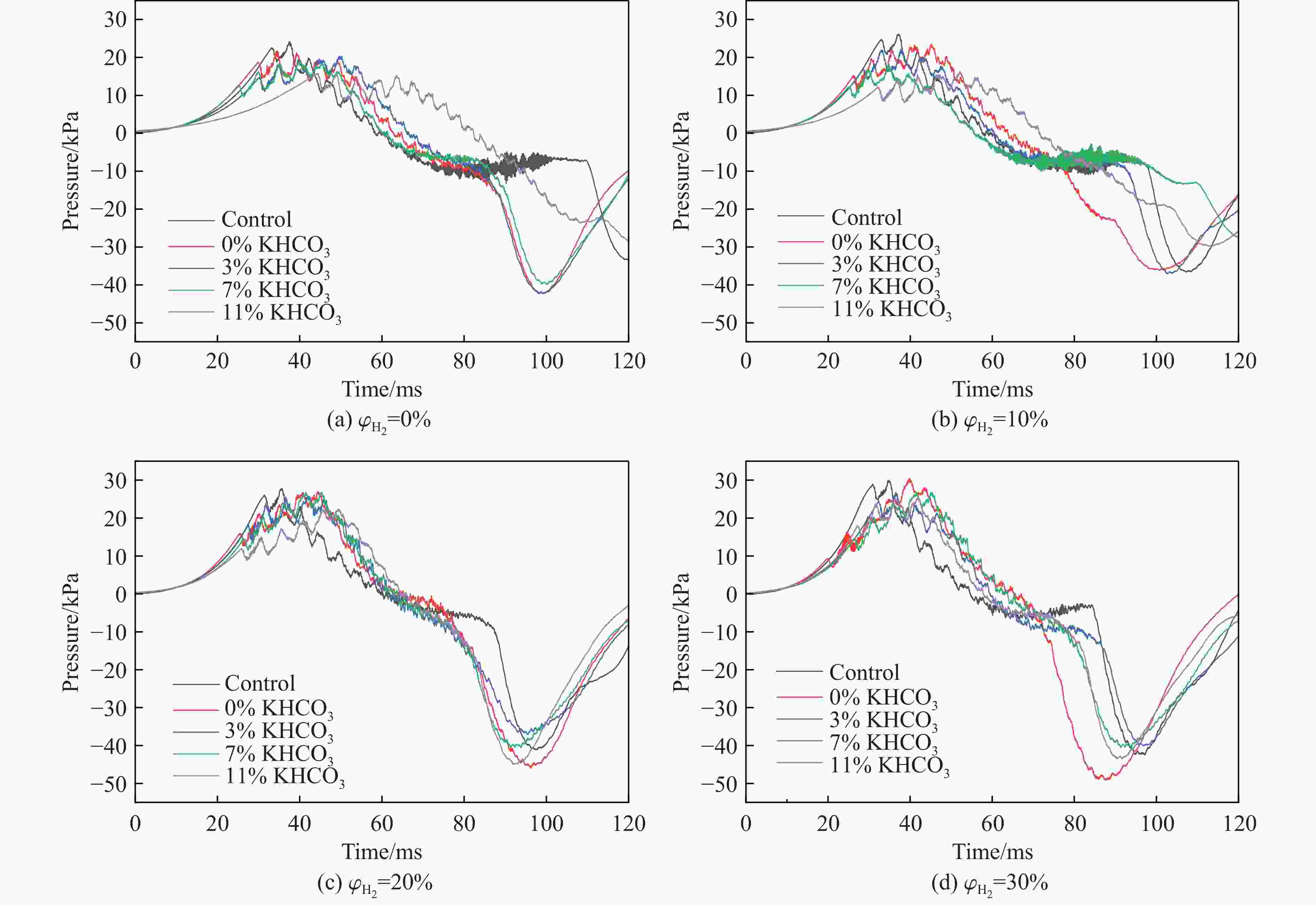
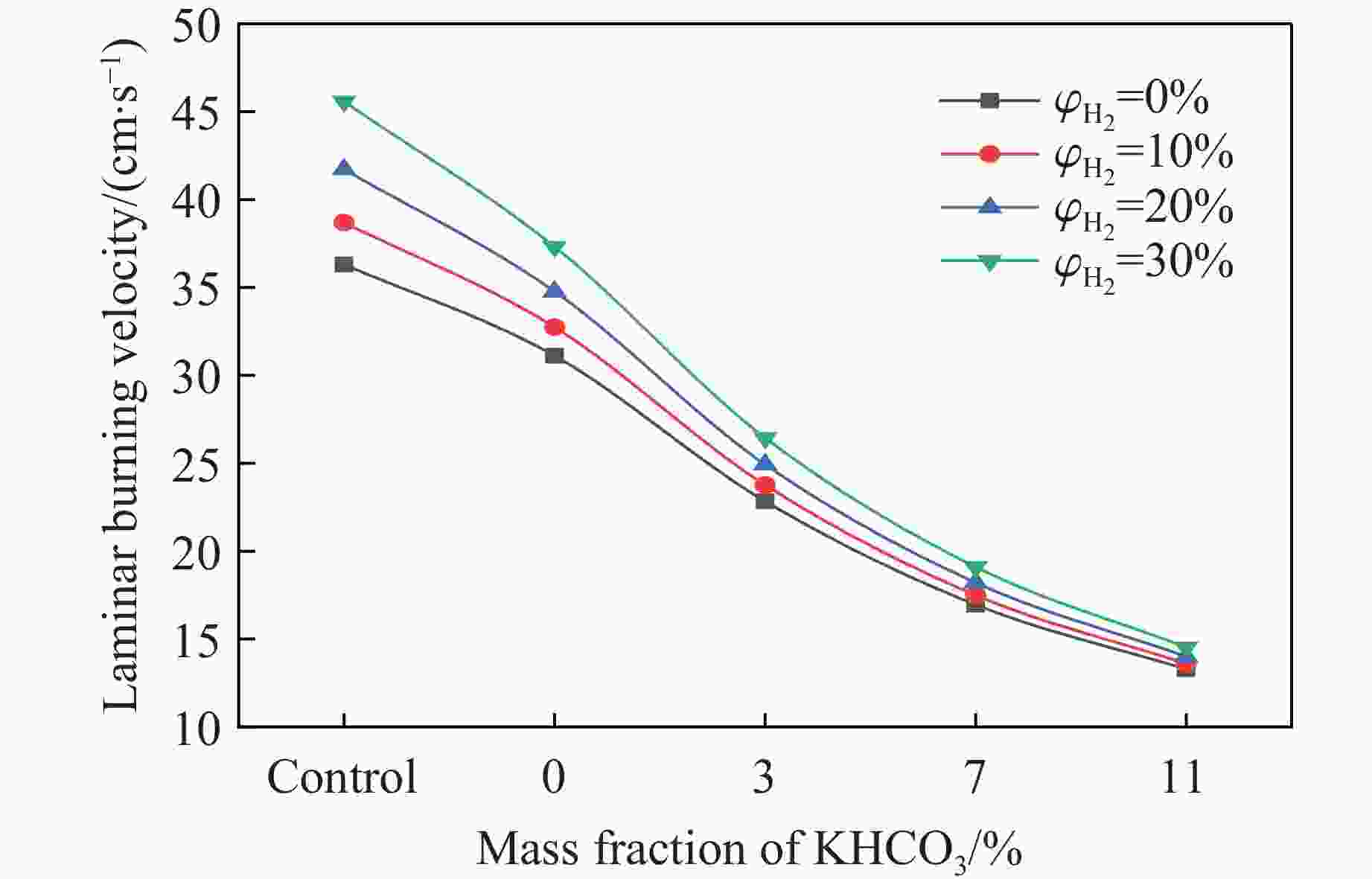
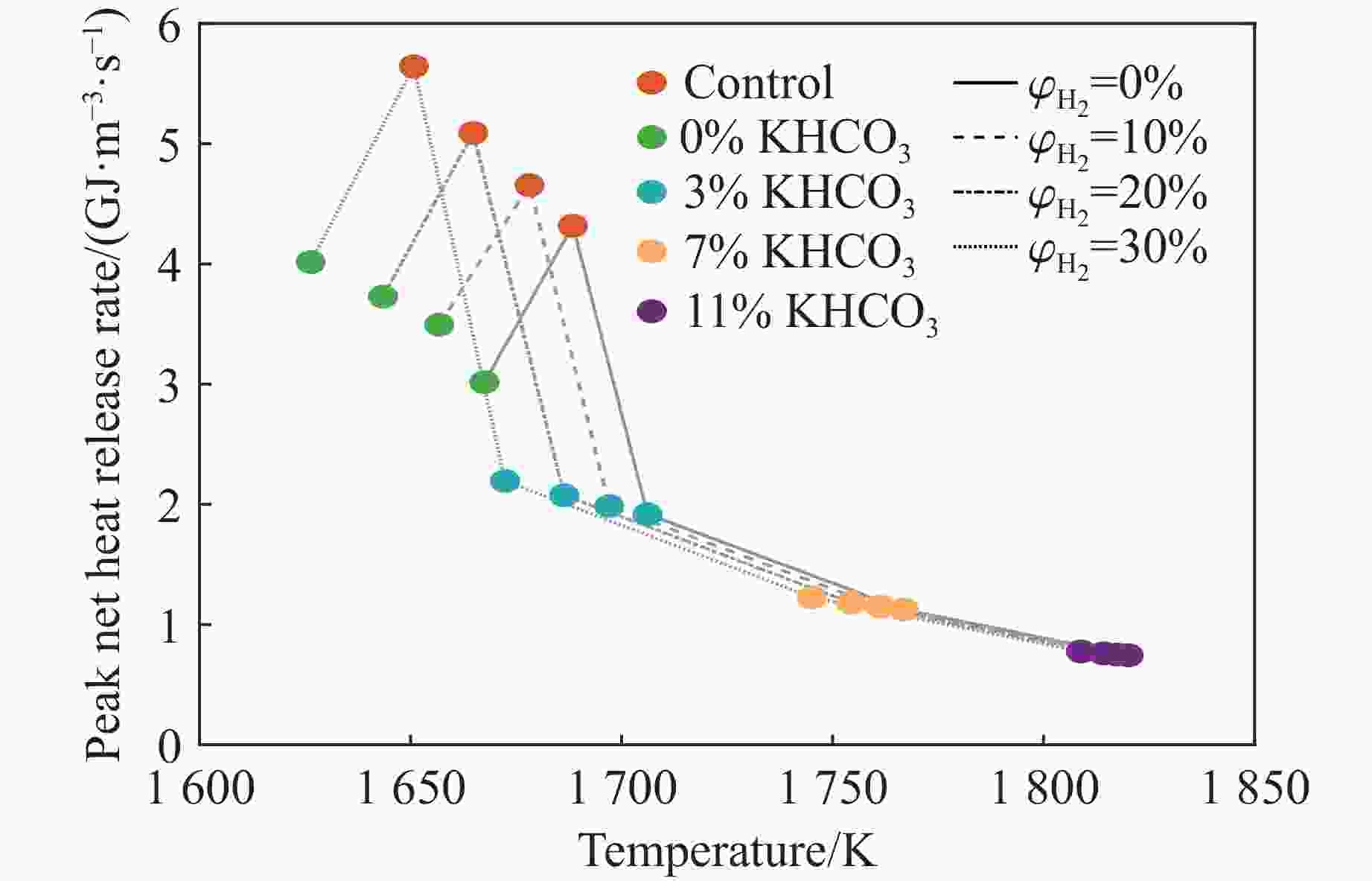
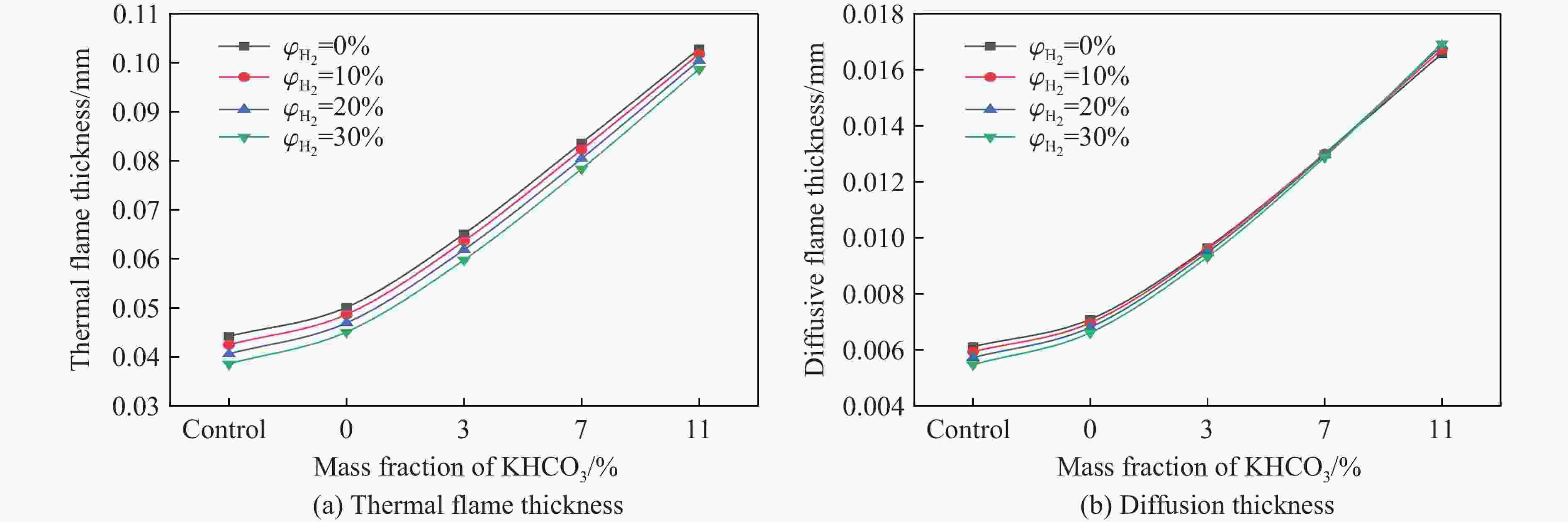
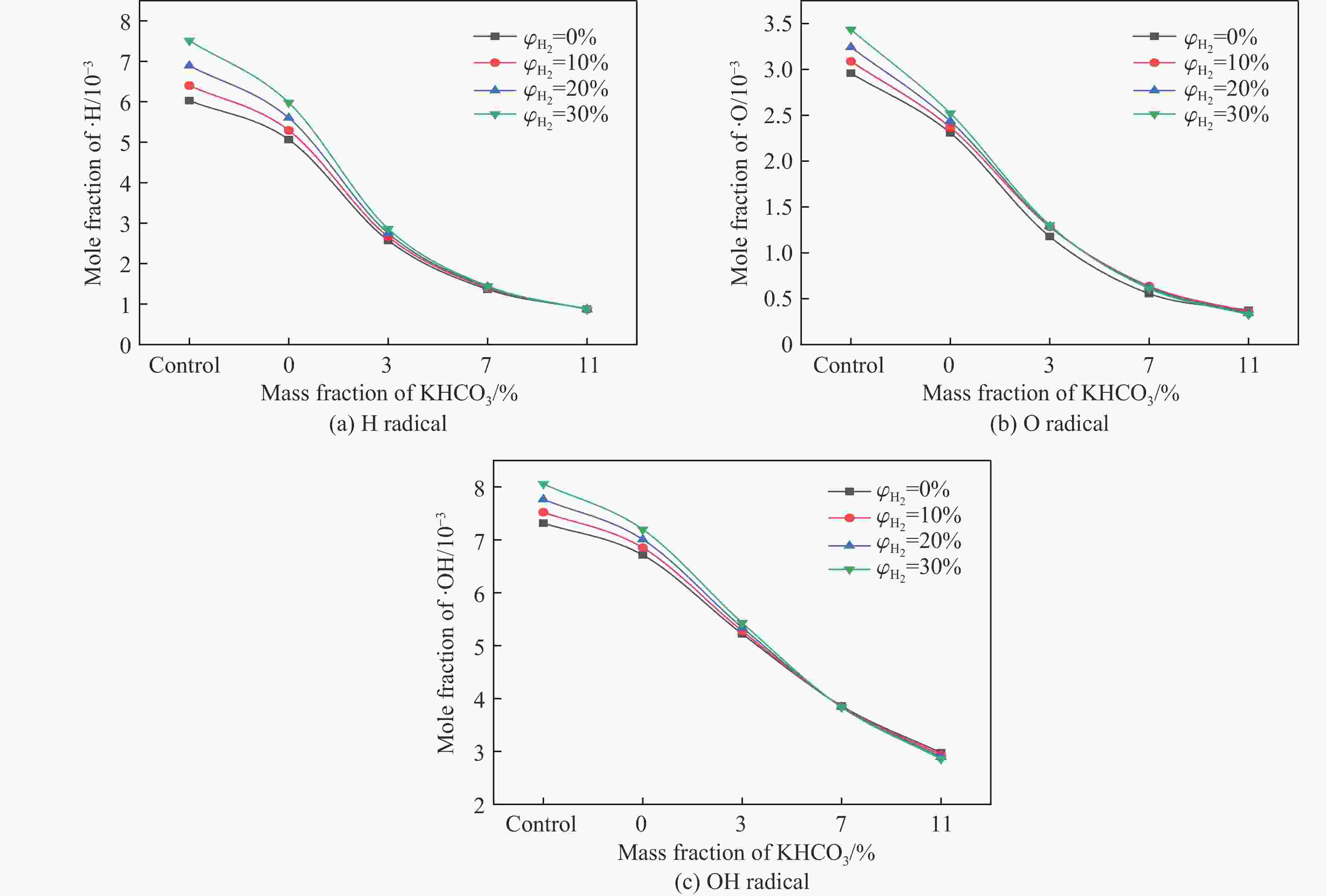
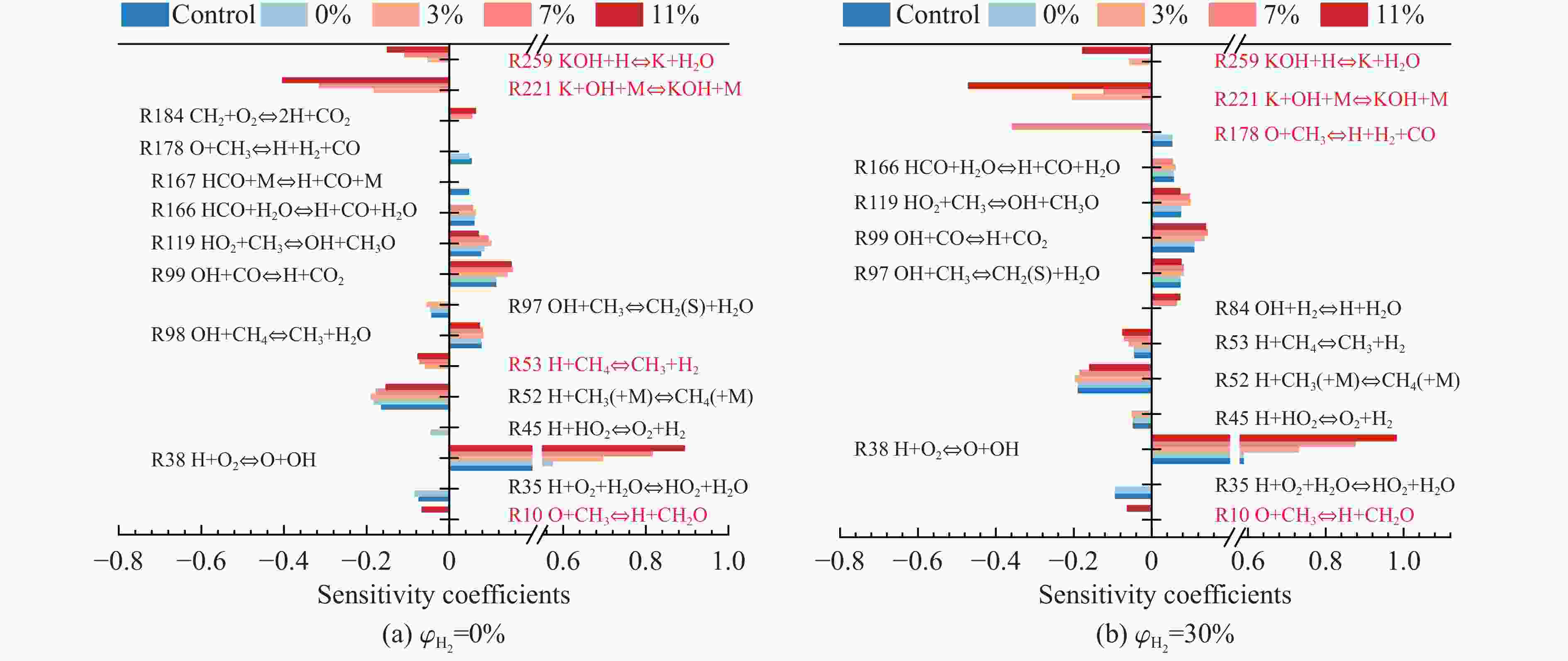
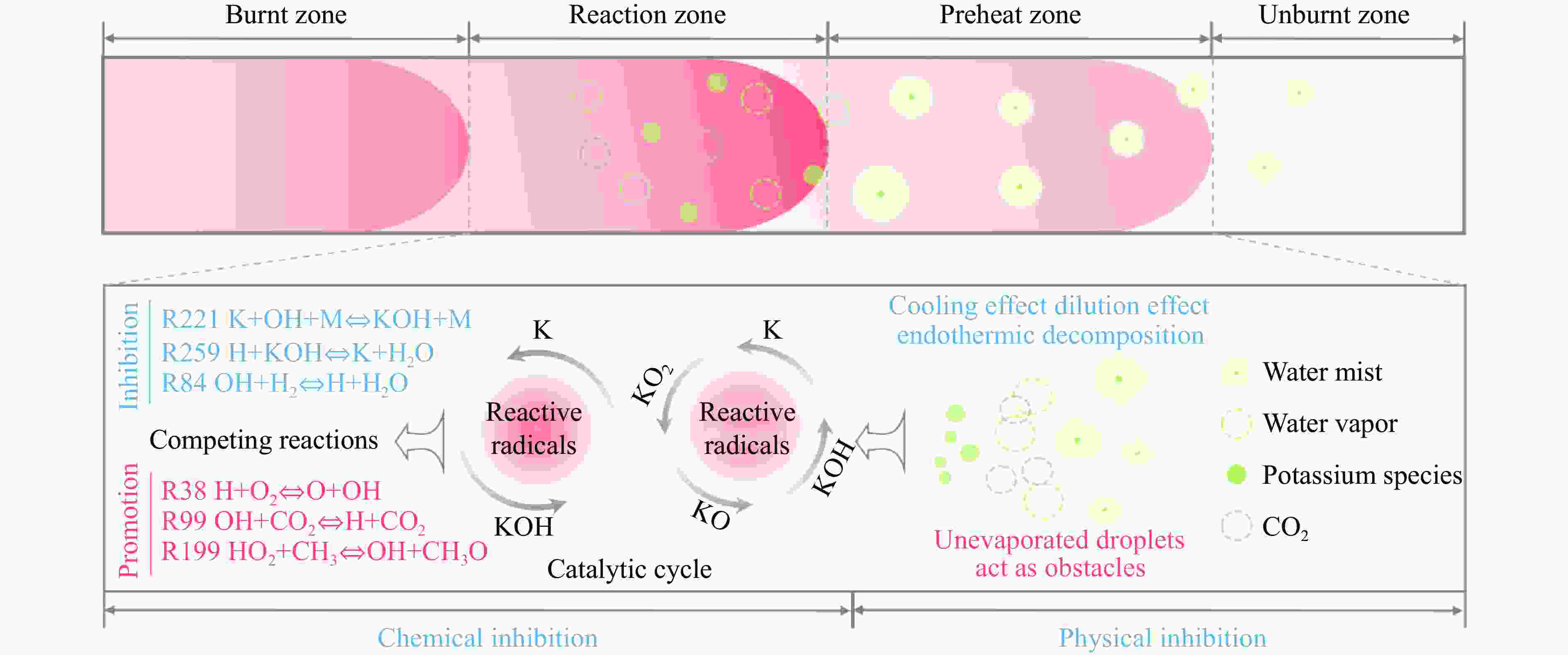
摘要: 甲烷-氢气混合气体的爆炸防控技术是保障氢能安全应用的重要课题。通过实验与数值模拟相结合的方法,系统研究了含KHCO3细水雾对甲烷-氢气预混爆燃的抑制机理。结果表明,含KHCO3细水雾对甲烷-氢气预混爆燃具有显著抑制效果,且抑制性能与KHCO3的质量分数呈正相关。以氢气体积分数为10%的混合气体为例,与对照组相比,KHCO3的质量分数为11%时,峰值压力和平均压力上升速率分别降低34.64%和44.57%,层流燃烧速度最高下降66.43%。KHCO3兼具物理与化学双重抑制效应:物理上,雾滴相变吸热和蒸气稀释效应降低火焰温度并稀释可燃物;化学上,KHCO3分解产生的钾化合物通过KOH→K→KOH重组循环消耗关键自由基(·H、·O、·OH),与链分支反应形成竞争,中断燃烧链式反应。此外,抑制过程是抑制与促进效应的竞争。高掺氢比和高KHCO3质量分数下,物理蒸发效率成为限制化学抑制作用的瓶颈,导致抑制效率出现饱和现象,但整体上仍表现出显著的抑制效果。Abstract: Explosion prevention and mitigation technologies for hydrogen-methane gas mixtures represent a critical research area for ensuring the safe application of hydrogen energy. This study systematically investigates the inhibition mechanism of potassium bicarbonate (KHCO3)-containing fine water mist on methane-hydrogen premixed deflagration using a combined approach of experiment and numerical simulation. The results indicate that KHCO3-containing fine water mist exhibits a significant inhibitory effect on methane-hydrogen premixed deflagration, with its suppression performance positively correlated to the KHCO3 mass fraction. Taking the condition of H2 volume fraction of 10% as an example, 11% KHCO3 addition resulted in reductions of the maximum explosion pressure and the average pressure rise rate by 34.64% and 44.57%, respectively. The laminar burning velocity was reduced by up to 66.43%. KHCO3 contributes to suppression through both physical and chemical mechanisms. Physically, droplet phase change (evaporation) absorbs heat and the generated steam dilutes the fuel mixture, thereby lowering the flame temperature and reducing reactant concentrations. Chemically, the decomposition of KHCO3 generates potassium compounds, which undergo the KOH→K→KOH recombination cycle to scavenge key radicals (·H, ·O, ·OH). This process competes with chain-branching reactions and interrupts the combustion chain reactions. Furthermore, the suppression process is governed by a competition between inhibitory and promotional effects. At high hydrogen blending ratios and high mass fractions of KHCO3, the physical evaporation efficiency becomes a bottleneck that constrains the chemical inhibition, leading to a saturation of the overall suppression efficiency. Nevertheless, a significant inhibitory effect is still maintained.
-
Key words:
- fine water mist /
- KHCO3 /
- methane-hydrogen /
- premixed deflagration /
- inhibition mechanism
-
表 1 含KHCO3细水雾对峰值压力和平均压力上升速率的影响
Table 1. Effects of KHCO3-containing fine water mist on the maximum explosion pressure and the average rate of pressure rise
$ {\varphi}_{{{\mathrm{H}}_{2}}} $/% Mass fraction of KHCO3/% $ {t}_{{{\mathrm{p}}_{\max }}} $/ms pmax/kPa $ {(\mathrm{d}p/\mathrm{d}t)}_{\text{avg}} $/(MPa·s−1) 0 Control 37.62 24.13±0.84 0.64±0.03 0 34.54 21.74±0.61 0.63±0.03 3 49.80 20.34±0.92 0.41±0.02 7 39.90 18.71±0.41 0.47±0.02 11 44.36 15.77±0.63 0.36±0.01 10 Control 37.18 26.16±0.81 0.70±0.03 0 45.14 23.46±0.59 0.52±0.02 3 37.70 22.16±0.95 0.59±0.02 7 34.82 18.31±0.73 0.53±0.02 11 52.12 16.29±0.34 0.31±0.01 20 Control 35.54 27.75±0.83 0.78±0.03 0 44.48 26.95±1.12 0.61±0.03 3 42.02 25.74±0.62 0.61±0.02 7 41.50 26.79±1.23 0.65±0.02 11 49.42 22.40±0.72 0.45±0.02 30 Control 34.88 29.92±0.78 0.86±0.03 0 39.82 30.49±1.49 0.77±0.03 3 36.52 26.72±0.88 0.73±0.02 7 41.50 26.79±0.75 0.65±0.03 11 41.84 25.36±1.07 0.61±0.02 -
[1] 孟庆强, 金之钧, 刘全有, 等. 天然氢气研究的现状、进展及展望 [J]. 石油与天然气地质, 2024, 45(5): 1483–1501. doi: 10.11743/ogg20240519MENG Q Q, JIN Z J, LIU Q Y, et al. Current status, advances, and prospects of research on natural hydrogen [J]. Oil & Gas Geology, 2024, 45(5): 1483–1501. doi: 10.11743/ogg20240519 [2] JOHNSON N, LIEBREICH M, KAMMEN D M, et al. Realistic roles for hydrogen in the future energy transition [J]. Nature Reviews Clean Technology, 2025, 1(5): 351–371. doi: 10.1038/s44359-025-00050-4 [3] BORETTI A, POLLET B G. Hydrogen economy: paving the path to a sustainable, low-carbon future [J]. International Journal of Hydrogen Energy, 2024, 93: 307–319. doi: 10.1016/j.ijhydene.2024.10.350 [4] 刘虎, 李权, 吕兆文, 等. 圆柱形障碍物对2H2+O2+nAr预混气体的再起爆实验研究 [J]. 高压物理学报, 2023, 37(5): 055202. doi: 10.11858/gywlxb.20230672LIU H, LI Q, LYU Z W, et al. Experimental study on re-initiation of 2H2+O2+nAr premixed gas by cylindrical obstacle [J]. Chinese Journal of High Pressure Physics, 2023, 37(5): 055202. doi: 10.11858/gywlxb.20230672 [5] 马秋菊, 邵俊程, 王众山, 等. 氢气比例和点火能量对CH4-H2混合气体爆炸强度影响的实验研究 [J]. 高压物理学报, 2020, 34(1): 015201. doi: 10.11858/gywlxb.20190803MA Q J, SHAO J C, WANG Z S, et al. Experimental study of the hydrogen proportion and ignition energy effects on the CH4-H2 mixture explosion intensity [J]. Chinese Journal of High Pressure Physics, 2020, 34(1): 015201. doi: 10.11858/gywlxb.20190803 [6] WANG Z J, GOU X L, ZHANG H W. Explosion limit of hydrogen/oxygen mixture with water vapor addition [J]. International Journal of Hydrogen Energy, 2024, 50: 772–781. doi: 10.1016/J.IJHYDENE.2023.06.321 [7] WEI S N, DENG H X, XU Z Z, et al. Effects of CO2 addition on deflagration characteristics of syngas-air premixed mixtures in T-pipeline [J]. International Journal of Hydrogen Energy, 2022, 47(87): 37125–37137. doi: 10.1016/j.ijhydene.2022.08.272 [8] LIU Q Q, LIU L Q, LIU Z Y, et al. Effects of H2 blended ratio and N2/CO2 dilution fraction on the deflagration shock wave of H2NG in slender closed pipelines [J]. International Journal of Hydrogen Energy, 2024, 73: 451–461. doi: 10.1016/j.ijhydene.2024.06.072 [9] LIANG Z H, ZHANG B, YUE W Y, et al. Suppression effects of PA@SH-SiC porous materials on hydrogen-air deflagration [J]. Journal of Loss Prevention in the Process Industries, 2025, 96: 105664. doi: 10.1016/j.jlp.2025.105664 [10] 计新龙, 杨克, 康青春, 等. 改性草木灰粉体抑制瓦斯爆炸的试验研究 [J]. 消防科学与技术, 2025, 44(6): 738–744. doi: 10.20168/j.1009-0029.2025.06.0738.07JI X L, YANG K, KANG Q C, et al. Experimental study on the inhibition of gas explosion by modified plant ash powder [J]. Fire Science and Technology, 2025, 44(6): 738–744. doi: 10.20168/j.1009-0029.2025.06.0738.07 [11] 裴蓓, 胡紫维, 韩谕良, 等. 含改性氯化合物对N2/细水雾抑制LPG爆炸影响研究 [J]. 爆炸与冲击, 2024, 44(11): 115401. doi: 10.11883/bzycj-2023-0340PEI B, HU Z W, HAN Y L, et al. Study on influence of modified chlorine-containing compounds on N2/water mist to suppress LPG explosion [J]. Explosion and Shock Waves, 2024, 44(11): 115401. doi: 10.11883/bzycj-2023-0340 [12] 夏远辰, 张彬, 王博乔, 等. 超细水雾对氢气-甲烷预混气体爆燃抑制机理的实验研究 [J]. 大连海事大学学报, 2022, 48(4): 127–134. doi: 10.16411/j.cnki.issn1006-7736.2022.04.015XIA Y C, ZHANG B, WANG B Q, et al. Experimental research on suppression mechanism of ultrafine water mist on deflagration of hydrogen-methane premixed gas [J]. Journal of Dalian Maritime University, 2022, 48(4): 127–134. doi: 10.16411/j.cnki.issn1006-7736.2022.04.015 [13] FRIEDMAN R, LEVY J B. Inhibition of opposed jet methane-air diffusion flames. the effects of alkali metal vapours and organic halides [J]. Combustion and Flame, 1963, 7: 195–201. doi: 10.1016/0010-2180(63)90179-2 [14] ZHANG T W, LIU H, HAN Z Y, et al. Numerical model for the chemical kinetics of potassium species in methane/air cup-burner flames [J]. Energy & Fuels, 2017, 31(4): 4520–4530. doi: 10.1021/acs.energyfuels.7b00106 [15] 王晓玲, 刘震起. 甲烷-空气预混区外含钾细水雾抑爆特性研究 [J]. 中国安全科学学报, 2024, 34(1): 150–157. doi: 10.16265/j.cnki.issn1003-3033.2024.01.0695WANG X L, LIU Z Q. Study on explosion suppression characteristics of water mist containing potassium compounds outside methane-air premixed area [J]. China Safety Science Journal, 2024, 34(1): 150–157. doi: 10.16265/j.cnki.issn1003-3033.2024.01.0695 [16] PEI B, LI S L, YANG S J, et al. Flame propagation inhibition study on methane/air explosion using CO2 twin-fluid water mist containing potassium salt additives [J]. Journal of Loss Prevention in the Process Industries, 2022, 78: 104817. doi: 10.1016/j.jlp.2022.104817 [17] HU Z W, PEI B, XU M J, et al. Study on the inhibition effect and mechanism of N2 twin-fluid water mist with modified chloride compounds on LPG explosion [J]. Energy, 2024, 291: 130394. doi: 10.1016/j.energy.2024.130394 [18] JIA J Z, TIAN X Y, WANG F X. Study on the effect of KHCO3 particle size and powder spraying pressure on the methane explosion suppression characteristics of pipe networks [J]. ACS Omega, 2022, 7(36): 31974–31982. doi: 10.1021/acsomega.2c02945 [19] VAN WINGERDEN K, WILKINS B. The influence of water sprays on gas explosions. part 1: water-spray-generated turbulence [J]. Journal of Loss Prevention in the Process Industries, 1995, 8(2): 53–59. doi: 10.1016/0950-4230(95)00002-I [20] CHEIKHRAVAT H, GOULIER J, BENTAIB A, et al. Effects of water sprays on flame propagation in hydrogen/air/steam mixtures [J]. Proceedings of the Combustion Institute, 2015, 35(3): 2715–2722. doi: 10.1016/j.proci.2014.05.102 [21] ZHANG P P, ZHOU Y H, CAO X Y, et al. Enhancement effects of methane/air explosion caused by water spraying in a sealed vessel [J]. Journal of Loss Prevention in the Process Industries, 2014, 29: 313–318. doi: 10.1016/j.jlp.2014.03.014 [22] INGRAM J M, AVERILL A F, BATTERSBY P, et al. Suppression of hydrogen/oxygen/nitrogen explosions by fine water mist containing sodium hydroxide additive [J]. International Journal of Hydrogen Energy, 2013, 38(19): 8002–8010. doi: 10.1016/j.ijhydene.2013.04.048 [23] SHAO P, MI H F, LUO N, et al. Characteristic behavior of methane/hydrogen premixed flame in ultrafine water mist with potassium additives [J]. Combustion Science and Technology, 2025, 197(12): 2981–2996. doi: 10.1080/00102202.2024.2327604 [24] BABUSHOK V I, LINTERIS G T, HOORELBEKE P, et al. Flame inhibition by potassium-containing compounds [J]. Combustion Science and Technology, 2017, 189(12): 2039–2055. doi: 10.1080/00102202.2017.1347162 [25] XIAO H H, SUN J H, CHEN P. Experimental and numerical study of premixed hydrogen/air flame propagating in a combustion chamber [J]. Journal of Hazardous Materials, 2014, 268: 132–139. doi: 10.1016/j.jhazmat.2013.12.060 [26] CAO X Y, REN J J, BI M S, et al. Experimental research on the characteristics of methane/air explosion affected by ultrafine water mist [J]. Journal of Hazardous Materials, 2017, 324: 489–497. doi: 10.1016/j.jhazmat.2016.11.017 [27] BERGER L, GRINBERG M, JÜRGENS B, et al. Flame fingers and interactions of hydrodynamic and thermodiffusive instabilities in laminar lean hydrogen flames [J]. Proceedings of the Combustion Institute, 2023, 39(2): 1525–1534. doi: 10.1016/j.proci.2022.07.010 [28] CHAKRAVARTHY S R, SAMPATH R, RAMANAN V. Dynamics and diagnostics of flame-acoustic interactions [J]. Combustion Science and Technology, 2017, 189(3): 395–437. doi: 10.1080/00102202.2016.1202938 [29] O’CONNOR J. Understanding the role of flow dynamics in thermoacoustic combustion instability [J]. Proceedings of the Combustion Institute, 2023, 39(4): 4583–4610. doi: 10.1016/j.proci.2022.07.115 [30] ZHENG R, BRAY K N C, ROGG B. Effect of sprays of water and NaCl-water solution on the extinction of laminar premixed methane-air counterflow flames [J]. Combustion Science and Technology, 1997, 126(1): 389–401. [31] 吕森林, 胡二江, 殷阁媛, 等. 稀释剂对氢气层流燃烧速度影响的实验和数值研究 [J]. 工程热物理学报, 2025, 46(7): 2427–2434.LYU S L, HU E J, YIN G Y, et al. Experimental and numerical study on the effect of diluent on laminar flame speeds of hydrogen [J]. Journal of Engineering Thermophysics, 2025, 46(7): 2427–2434. [32] FAN R J, WANG Z R, GUO W J, et al. Experimental and theoretical study on the suppression effect of CF3CHFCF3 (FM-200) on hydrogen-air explosion [J]. International Journal of Hydrogen Energy, 2022, 47(26): 13191–13198. doi: 10.1016/j.ijhydene.2022.02.062 [33] 李慧真. 氨气掺混二甲醚/氢气基础燃爆特性及动力学机制研究 [D]. 合肥: 中国科学技术大学, 2024.LI H Z. Study on the fundamental characteristics and mechanism of combustion and explosion of ammonia blended with dimethyl ether/hydrogen [D]. Hefei: University of Science and Technology of China, 2024. [34] CONCETTI R, HASSLBERGER J, CHAKRABORTY N, et al. Analysis of water droplet interaction with turbulent premixed and spray flames using carrier phase direct numerical simulations [J]. Combustion Science and Technology, 2023, 195(7): 1411–1433. doi: 10.1080/00102202.2023.2182192 [35] SEYFERT C, RODRÍGUEZ-RODRÍGUEZ J, LOHSE D, et al. Stability of respiratory-like droplets under evaporation [J]. Physical Review Fluids, 2022, 7(2): 023603. doi: 10.1103/PhysRevFluids.7.023603 [36] BADHUK P, RAVIKRISHNA R V. A numerical study on the response of chemically active flame inhibitors to strain rate variations [J]. Proceedings of the Combustion Institute, 2021, 38(3): 4615–4623. doi: 10.1016/j.proci.2020.05.023 [37] BADHUK P, RAVIKRISHNA R V. Flame inhibition by aqueous solution of alkali salts in methane and LPG laminar diffusion flames [J]. Fire Safety Journal, 2022, 130: 103586. doi: 10.1016/j.firesaf.2022.103586 [38] SLACK M, COX J W, GRILLO A, et al. Potassium kinetics in heavily seeded atmospheric pressure laminar methane flames [J]. Combustion and Flame, 1989, 77(3/4): 311–320. doi: 10.1016/0010-2180(89)90137-5 -






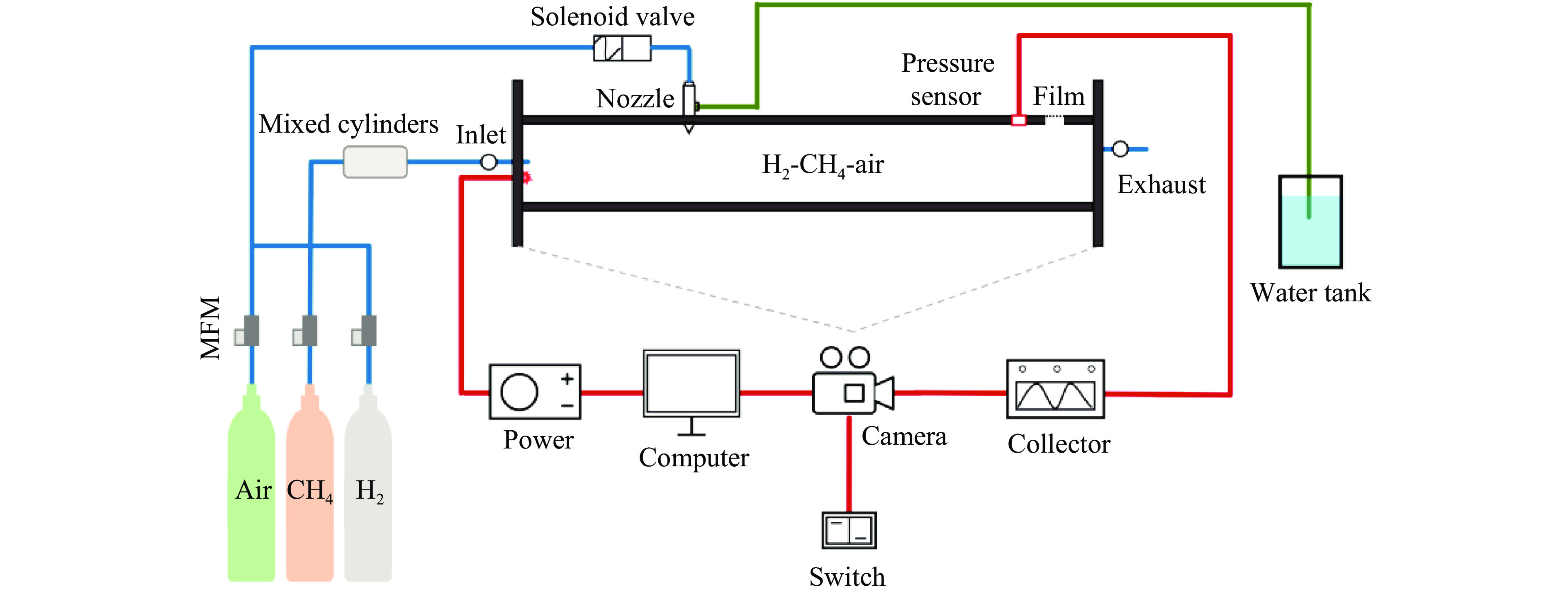
 下载:
下载: