Detonation Performance and Equation of State of Low Density Explosive Mixed by PBX-9502 Powder and 502 Glue
-
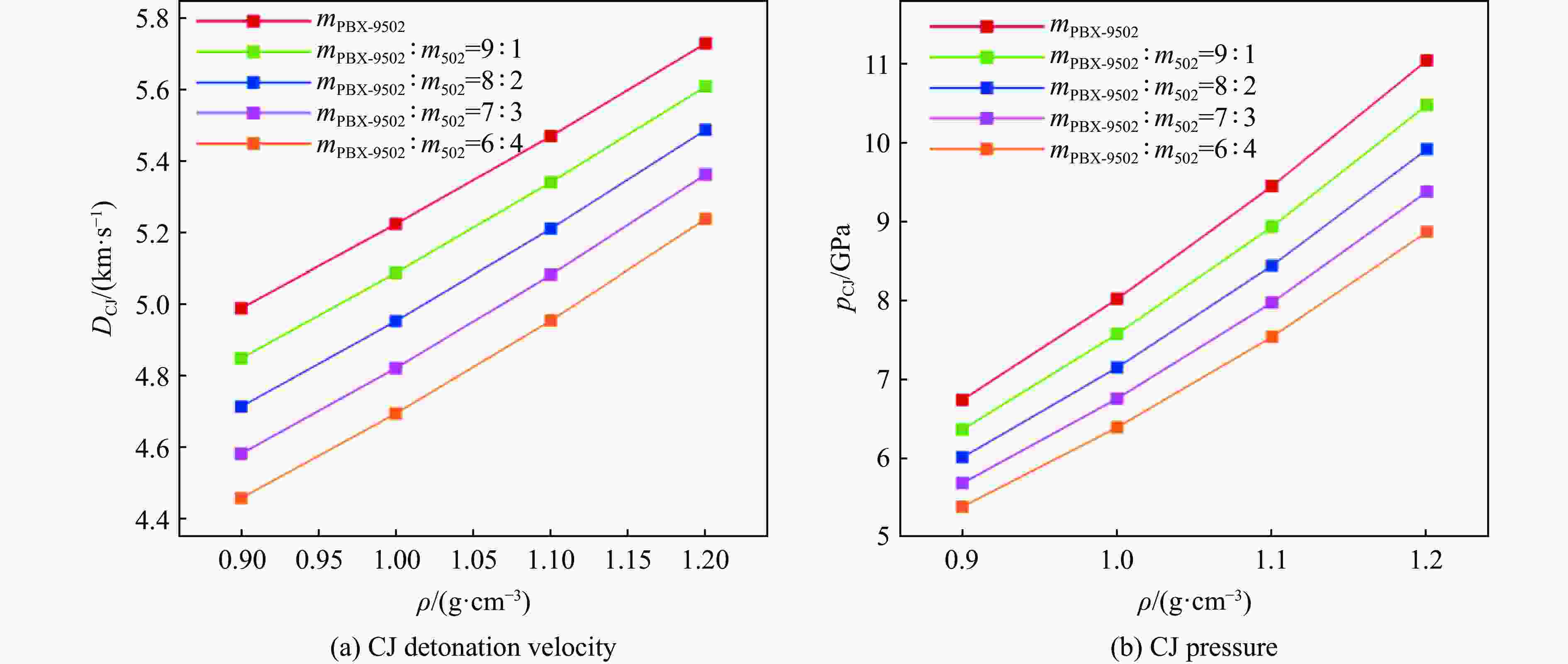
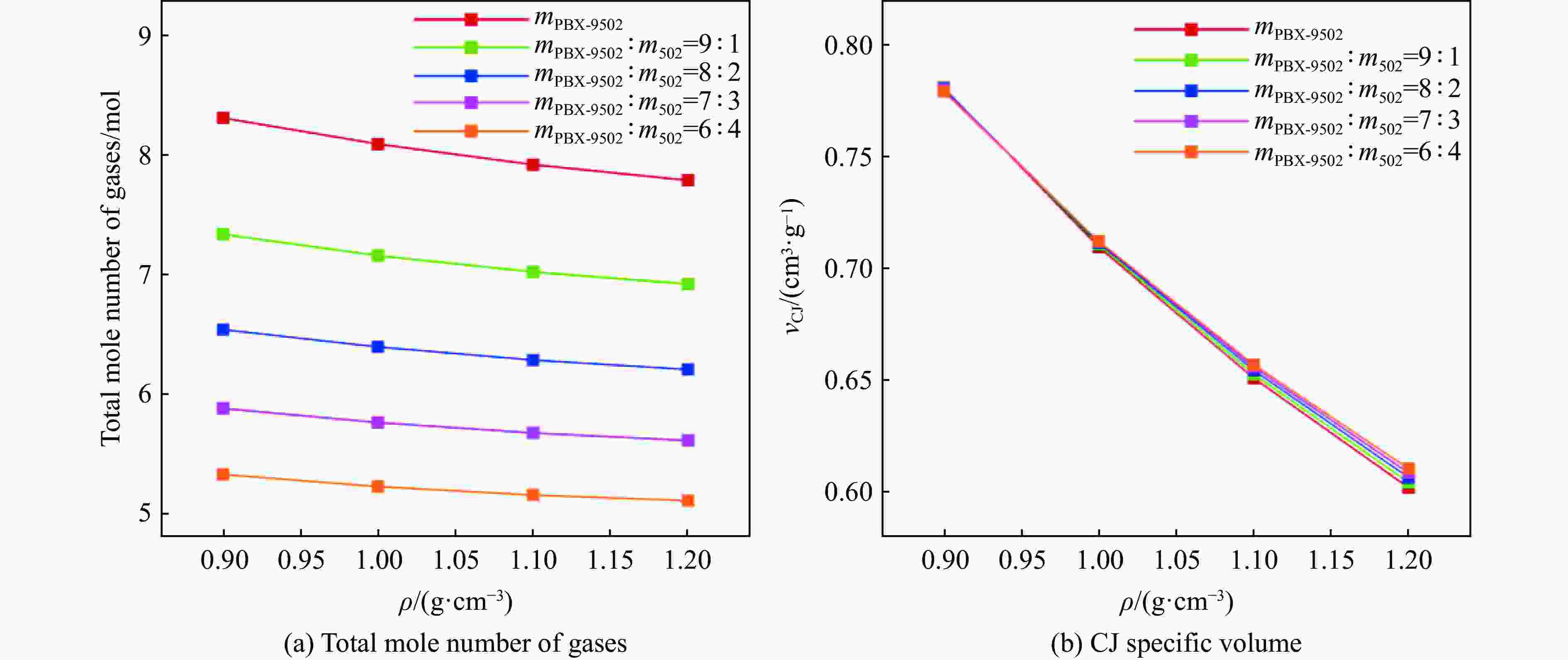
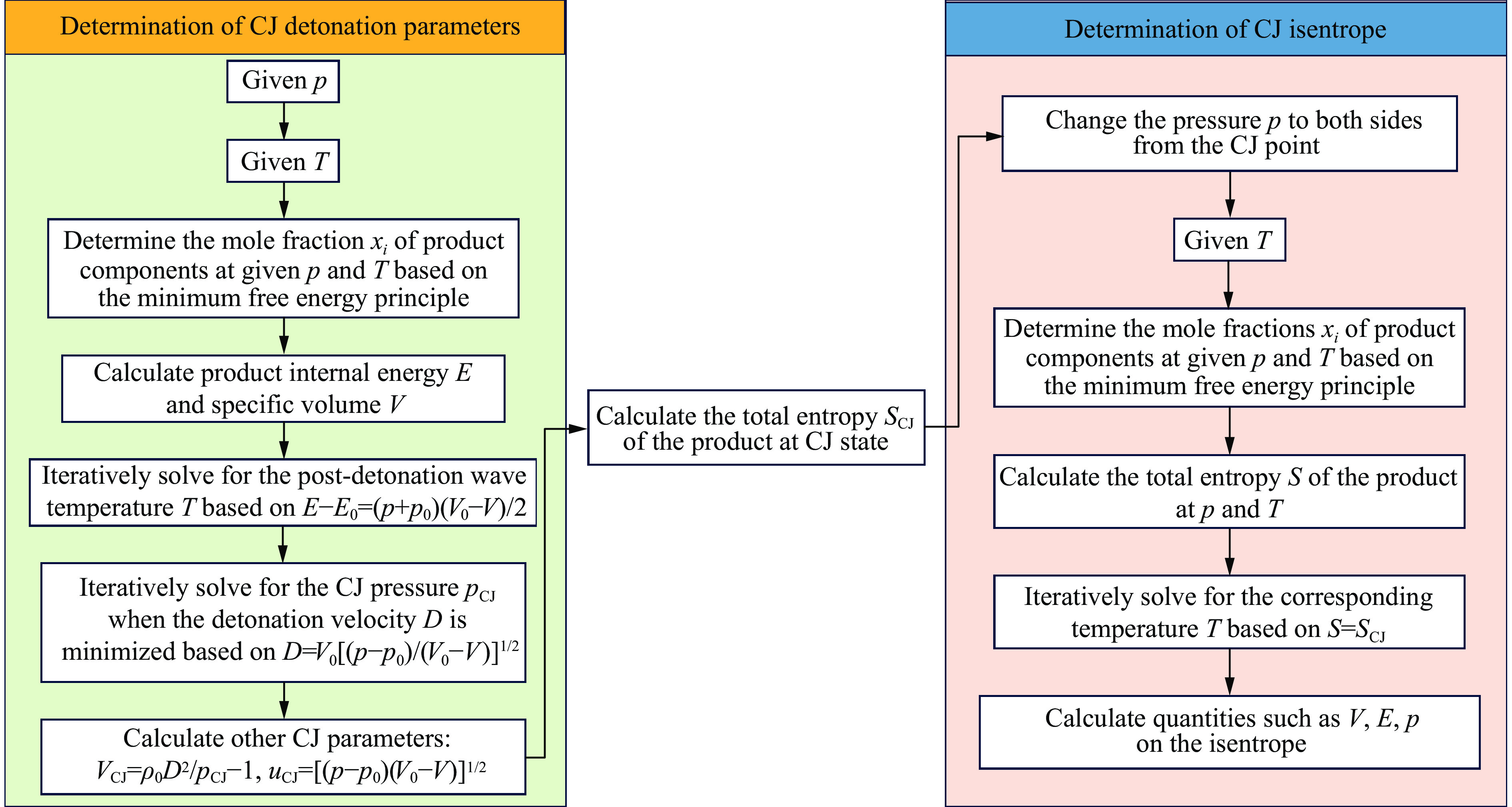
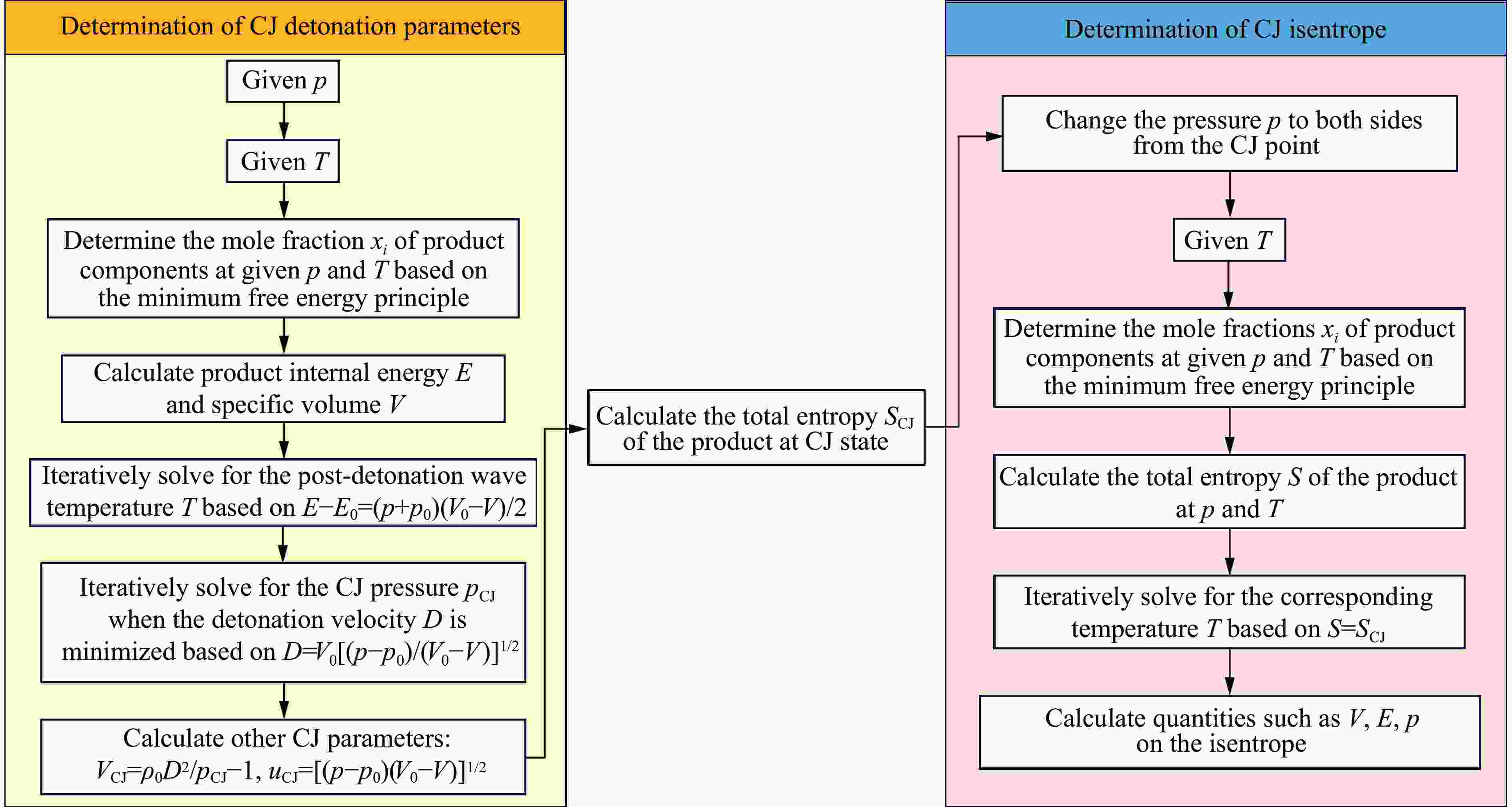
摘要: 针对PBX-9502粉末与502胶按一定比例混合的低密度炸药,基于BKW程序,采用理论分析方法,对其爆轰性能和状态方程(equation of state,EOS)进行了研究。采用基团贡献法,计算了Kel-F(PBX-9502黏结剂)和氰基丙烯酸乙酯(502胶主要成分)的生成热,确定了18种含F、Cl元素的产物气体的标准熵温度系数和余容,在BKW程序框架下,针对5种配比、4种密度状态给出了爆轰产物的CJ爆速和爆压,并根据计算所得CJ等熵线拟合出相应的JWL状态方程参数。结果表明,爆速、爆压与初始密度正相关,而与502胶含量负相关。所得JWL状态方程参数可供通用爆轰计算软件使用,以实现装置爆轰性能评估,为实际应用中502胶配比和混合装药密度选择提供了理论依据,相关方法也可直接推广至其他配方炸药(硫、铝)的爆轰参数研究中,具有重要的工程应用价值。Abstract: This study focuses on the detonation performance and equation of state (EOS) of a low-density composite explosive mixed in a certain proportion of PBX-9502 powder and 502 glue. The theoretical analysis method was used based on the BKW program. The main work includes calculating the heat of formation of Kel-F (PBX-9502 binder) and ethyl cyanoacrylate (main component of 502 glue) using the group contribution method, determining the standard entropy temperature coefficient and excess volume of 18 product gases containing F and Cl elements. Within the BKW program framework, the CJ detonation velocity and pressure were determined for five mixing ratios and four density states. The corresponding JWL equation of state parameters are fitted based on the calculated CJ isentropic line. The results show that the detonation velocity and pressure are positively correlated with initial density, but negatively correlated with the content of 502 glue. The relevant results provide a theoretical basis for the selection of 502 glue ratio and mixed charge density in practical applications. The obtained JWL EOS parameters can also be used by general detonation calculation software to evaluate the detonation performance of devices. The relevant method can also be directly extended to the study of detonation parameters of other formulated explosives (sulfur, aluminum), which has important engineering application value.
-
Key words:
- composite explosive /
- BKW /
- detonation parameter /
- JWL equation of state
-
Explosives α β θ κ Nitramines 0.5 0.16 400 10.90978 Nitroaromatics 0.5 0.09585 400 12.685 Triazoles 0.5 0.18 1 850 11.800 表 2 PBX-9502粉末与502胶在不同质量比下的元素计量与生成热
Table 2. Molecular formula and heat of formation of PBX-9502 powder and 502 adhesive at different mass ratios
No. wPBX/% w502/% Molecular formula Molecular
weight$ \Delta{H}_{\text{f}}^{\Theta} $ (298 K)/
(kJ·mol−1)C H N O F Cl 1 100 0 6.0636 5.8727 5.8091 5.8091 0.3500 0.0955 263.1088 − 204.5262 2 90 10 6.0516 6.0862 4.8983 5.0877 0.2837 0.0774 236.9762 − 219.5778 3 80 20 6.0417 6.2611 4.1521 4.4966 0.2294 0.0626 215.5657 − 231.9095 4 70 30 6.0335 6.4071 3.5295 4.0035 0.1841 0.0502 197.7035 − 242.1976 5 60 40 6.0265 6.5307 3.0023 3.5859 0.1457 0.0397 182.5749 − 250.9112 6 50 50 6.0205 6.6367 2.5499 3.2276 0.1128 0.0308 169.5971 − 258.3860 表 3 Kel-F 800和氰基丙烯酸乙酯生成热的基团贡献[9–11]
Table 3. The group contribution for heat of formation of Kel-F 800 and ethyl cyanoacrylate[9–11]
Group Kel-F Ethyl cyanoacrylate $ \Delta H_{\text{f}}^{\Theta} $ (298 K)/(kJ·mol−1) –CH3 0 1 −46.0 –CH2– 1 2 −22.0 >C< 7 1 20.0 –COO– 0 1 −337.0 –CN 0 1 123.0 –F 11 0 −195.0 –Cl 3 0 −49.0 表 4 含卤元素气体产物的标准熵温度系数
Table 4. Temperature polynomial coefficients of standard entropy for gases containing halogen elements product
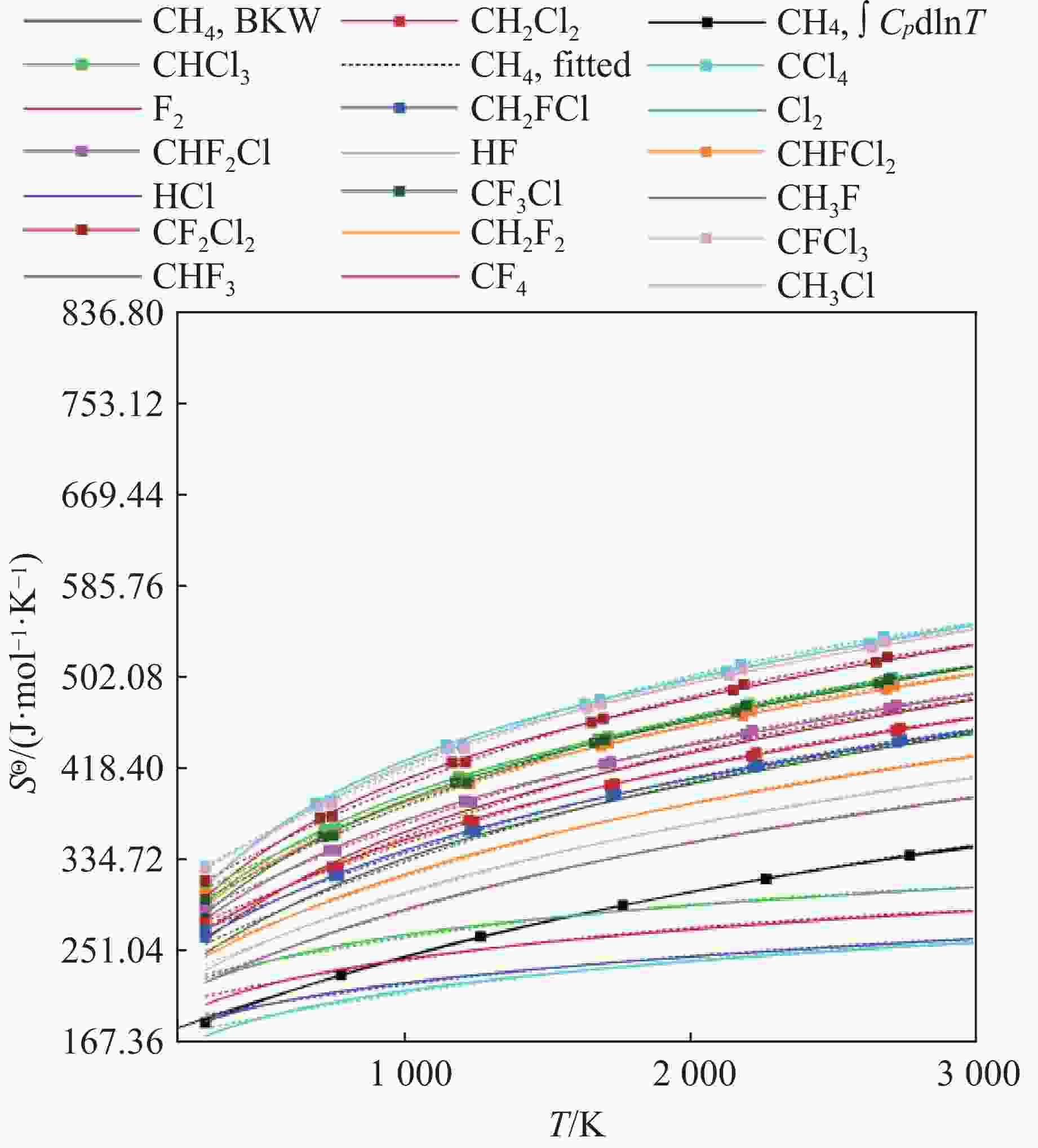
No. Product A0/(10 J·mol−1·K−1) A1/(10−2 J·mol−1·K−2) A2/(10−6 J·mol−1·K−3) A3/(10−10 J·mol−1·K−4) 1 F2 19.202864 6.376033 − 14.401663 13.141464 2 Cl2 21.250566 6.563334 − 15.080277 13.800264 3 HF 16.302011 6.375877 − 14.400920 13.140690 4 HCl 17.864983 5.294868 − 11.347577 10.431809 5 CH3F 19.491600 11.333699 − 21.022033 17.748069 6 CH2F2 21.704793 12.586229 − 24.495734 21.232386 7 CHF3 21.877056 14.124770 − 28.780668 25.679245 8 CF4 22.947865 15.777702 − 33.470989 30.495027 9 CH3Cl 20.696290 11.692847 − 21.893444 18.941069 10 CH2Cl2 24.141206 13.393581 − 26.766434 23.916686 11 CHCl3 26.593205 15.414378 − 32.774277 29.947137 12 CCl4 27.955831 17.717350 − 39.777178 36.912717 13 CH2FCl 23.518594 13.006725 − 25.655546 22.627426 14 CHF2Cl 25.027446 14.543305 − 30.076831 27.032850 15 CHFCl2 26.293778 15.037616 − 31.637213 28.717112 16 CF3Cl 25.420174 16.307227 − 35.161814 32.230888 17 CF2Cl2 26.952207 16.787842 − 36.702735 33.807209 18 CFCl3 27.913994 17.268819 − 38.276777 35.393522 表 5 含F、Cl元素的主要产物气体余容
Table 5. Covolumes of gases containing halogen elements
No. Gas product Covolume No. Gas product Covolume 1 F2 243.31 10 CH2Cl2 346.62 2 Cl2 301.69 11 CHCl3 410.37 3 HF 283.89 12 CCl4 443.86 4 HCl 254.42 13 CH2FCl 545.36 5 CH3F 506.25 14 CHF2Cl 694.77 6 CH2F2 602.89 15 CHFCl2 693.11 7 CHF3 839.15 16 CF3Cl 830.56 8 CF4 952.70 17 CF2Cl2 763.72 9 CH3Cl 351.62 18 CFCl3 853.15 表 6 不同密度PBX-9502炸药爆轰参数的BKW计算结果与文献数据对比
Table 6. Comparison of BKW calculation results and literature data for detonation parameters of PBX-9502 explosive with different densities
表 8 典型低密度炸药产物的JWL状态方程参数
Table 8. Parameters of JWL equation of state for product of typical low-density explosive
ρ/(g·cm−3) A/GPa B/GPa C/GPa R1 R2 ω 0.9 1828.260 54.243 2.414 14.24586 6.00797 0.91511 1.0 1859.854 55.895 2.785 12.86775 5.43208 0.91533 1.1 1919.808 60.710 3.172 11.82496 5.03780 0.91973 1.2 1947.197 65.817 3.571 10.92865 4.71671 0.92696 -
[1] MADER C L. Fortran BKW: a code for computing the detonation properties of explosives: LA-3704 [R]. Los Alamos: Los Alamos Scientific Laboratory of the University of California, 1967. [2] REE F H. A statistical mechanical theory of chemically reacting multiphase mixtures: application to the detonation properties of PETN [J]. The Journal of Chemical Physics, 1984, 81(3): 1251–1263. doi: 10.1063/1.447811 [3] FRIED L E. CHEETAH: a fast thermochemical code for detonation: UCRL-ID-115752 [R]. Livermore: Lawrence Livermore National Laboratory, 1993. [4] LI D W, ZHOU L, ZHANG X R. Partial reparametrization of the BKW equation of state for DNAN-based melt-cast explosives [J]. Propellants, Explosives, Pyrotechnics, 2017, 42(5): 499–505. doi: 10.1002/prep.201600206 [5] 赵艳红, 张弓木, 张其黎, 等. HMX炸药爆轰产物物态方程研究 [J]. 计算物理, 2017, 34(2): 155–159. doi: 10.3969/j.issn.1001-246X.2017.02.004ZHAO Y H, ZHANG G M, ZHANG Q L, et al. Equation of state of detonation products for HMX explosive [J]. Chinese Journal of Computational Physics, 2017, 34(2): 155–159. doi: 10.3969/j.issn.1001-246X.2017.02.004 [6] VAULLERIN M, ESPAGNACQ A, BLAISE B. Reparametrization of the BKW equation of state for the Triazoles and comparison of the detonation properties of HMX, TNMA and NTO by means of ab-initio and semiempirical calculations [J]. Propellants, Explosives, Pyrotechnics, 1998, 23(2): 73–76. doi: 10.1002/(SICI)1521-4087(199804)23:2<73::AID-PREP73>3.0.CO;2-M [7] KOZYREV N V. Reparametrization of the BKW equation of state for CHNO explosives which release no condensed carbon upon detonation [J]. Central European Journal of Energetic Materials, 2015, 12(4): 651–670. [8] SUCESKA M, CHAN H Y S, STIMAC B, et al. BKW EOS: history of modifications and further improvement of accuracy with temperature-dependent covolumes of polar molecules [J]. Propellants, Explosives, Pyrotechnics, 2023, 48(1): e202100278. doi: 10.1002/prep.202100278 [9] 董海山. 炸药及相关物性能 [M]. 绵阳: 中国工程物理研究院, 2005.Dong H S. Properties of explosives and related materials [M]. Mianyang: China Academy of Engineering Physics, 2005. [10] 黄整, 陈波, 刘福生. TATB生成焓的量子力学计算 [J]. 原子与分子物理学报, 2004, 21(3): 499–504. doi: 10.3969/j.issn.1000-0364.2004.03.027HUANG Z, CHEN B, LIU F S. Quantum calculation for the enthalpy of formation of TATB [J]. Journal of Atomic and Molecular Physics, 2004, 21(3): 499–504. doi: 10.3969/j.issn.1000-0364.2004.03.027 [11] VANKREVELEN D W. 聚合物的性质: 性质的估算及其与化学结构的关系 [M]. 许元泽, 赵锝禄, 吴大成, 译. 北京: 科学出版社, 1981.VANKREVELEN D W. Properties of polymers: their estimation and correlation with chemical structure [M]. Translated by XU Y Z, ZHAO D L, WU D C. Beijing: Science Press, 1981. [12] 国家标准局. NBS化学热力学性质表 SI的单位表示的无机物质和C1与C2有机物质选择值 [M]. 刘天和, 赵梦月, 译. 北京: 中国标准出版社, 1998.National Standards Administration. The NBS tables of chemical thermodynamic properties: selected values for inorganic and C1 and C2 organic substances in SI units [M]. Translated by LIU T H, ZHAO M Y. Beijing: Standards Press of China, 1998. [13] 马沛生, 高铭书, 江碧云, 等. 新近理想气体热容数据与温度的关联 [J]. 化工学报, 1979(2): 109–132.MA P S, GAO M S, JIANG B Y, et al. Correlation of the recent ideal gas heat capacity data with temperature [J]. Journal of Chemical Industry and Engineering, 1979(2): 109–132. [14] MADER C L. Detonation properties of condensed explosives computed using the Becker-Kistiakowsky-Wilson equation of state: LA-2900 [R]. Los Alamos: Los Alamos Scientific Laboratory of the University of California, 1963. [15] MENIKOFF R. SURFplus model calibration for PBX 9502: LA-UR-17-31015 [R]. Los Alamos: Los Alamos National Laboratory, 2017. [16] YU H, YU X, HUANG K B, et al. Numerical study of density effect on JB-9014 equation of state of detonation product [C]//Proceedings of the 46th International Annual Conference of Fraunholfer. ICT, 2015. -






 下载:
下载: