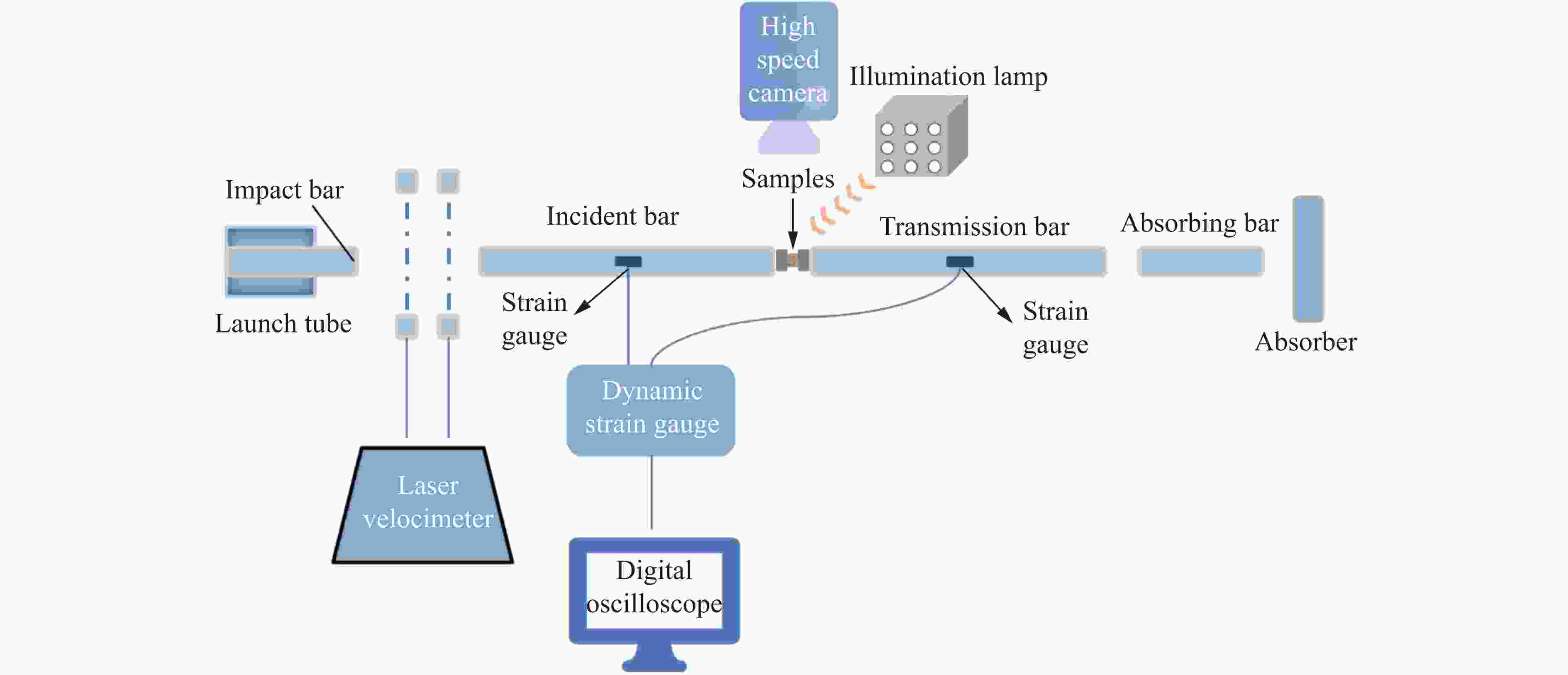
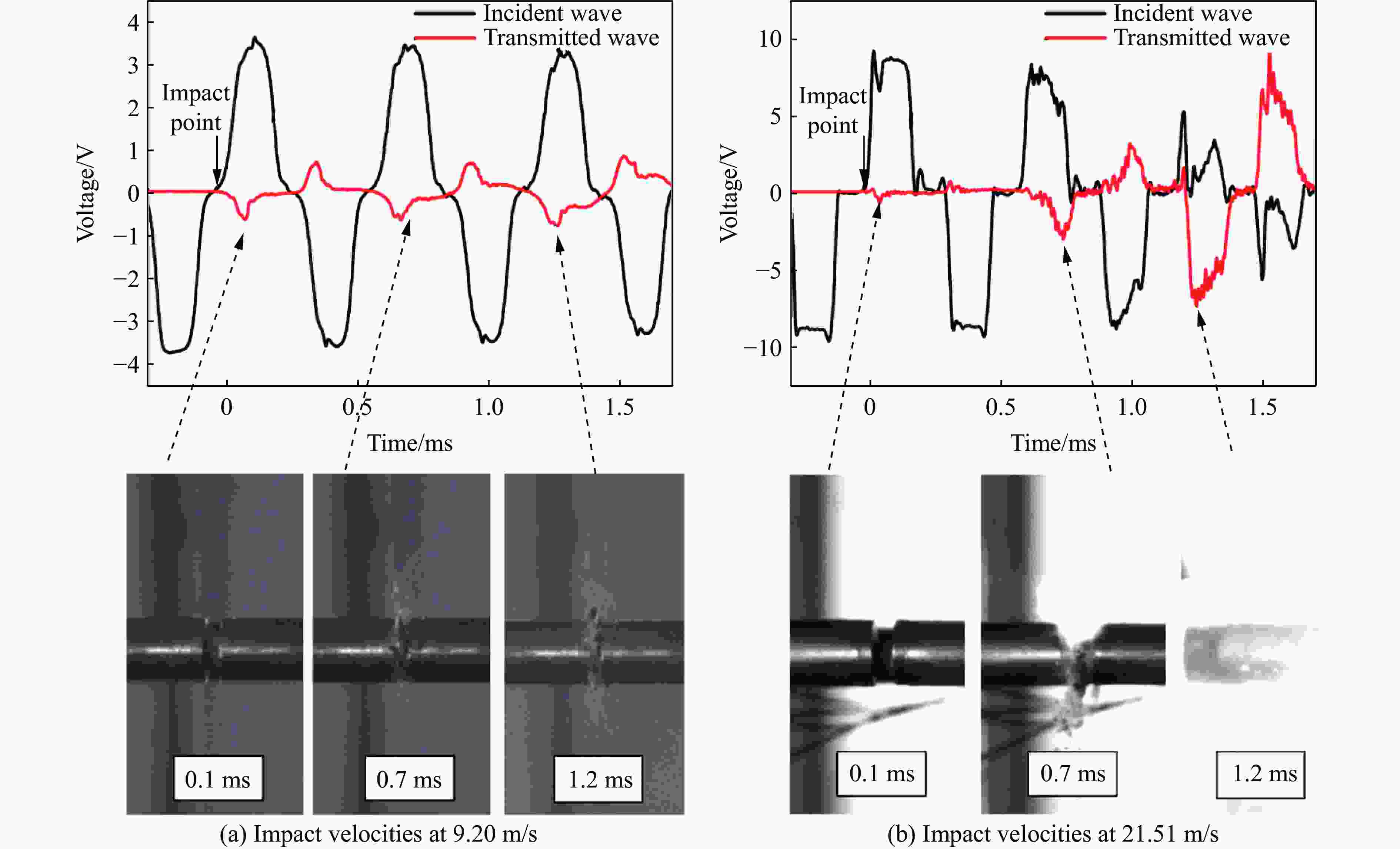
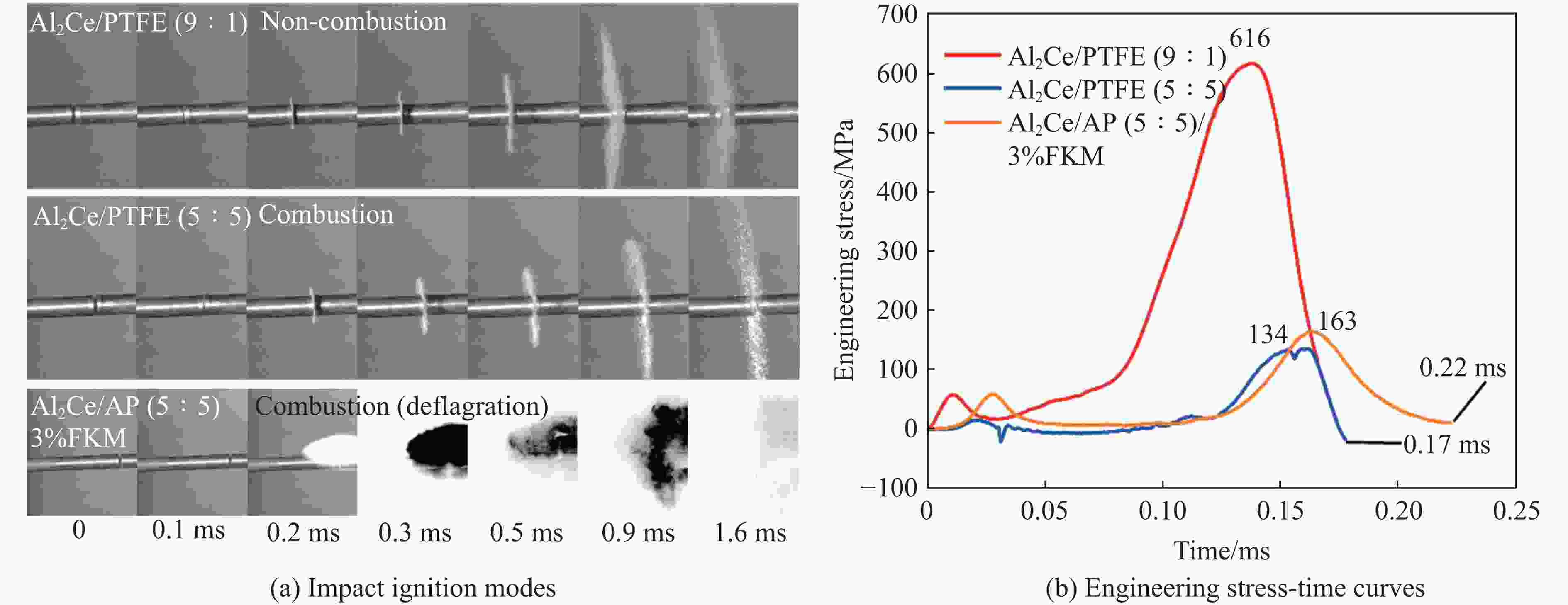
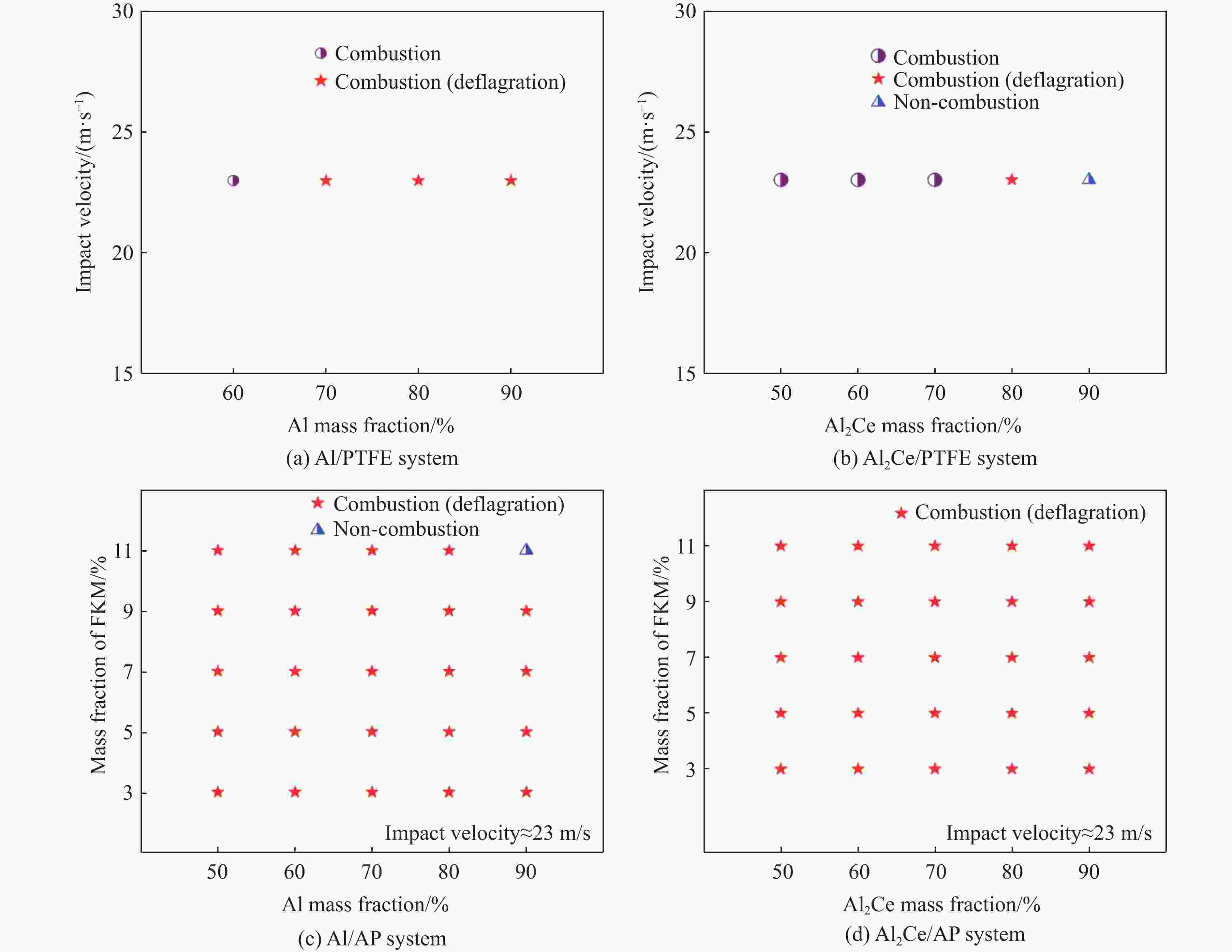
Mechanical Properties and Ignition Performance of Rare Earth Reactive Materials under Impact Loading
-
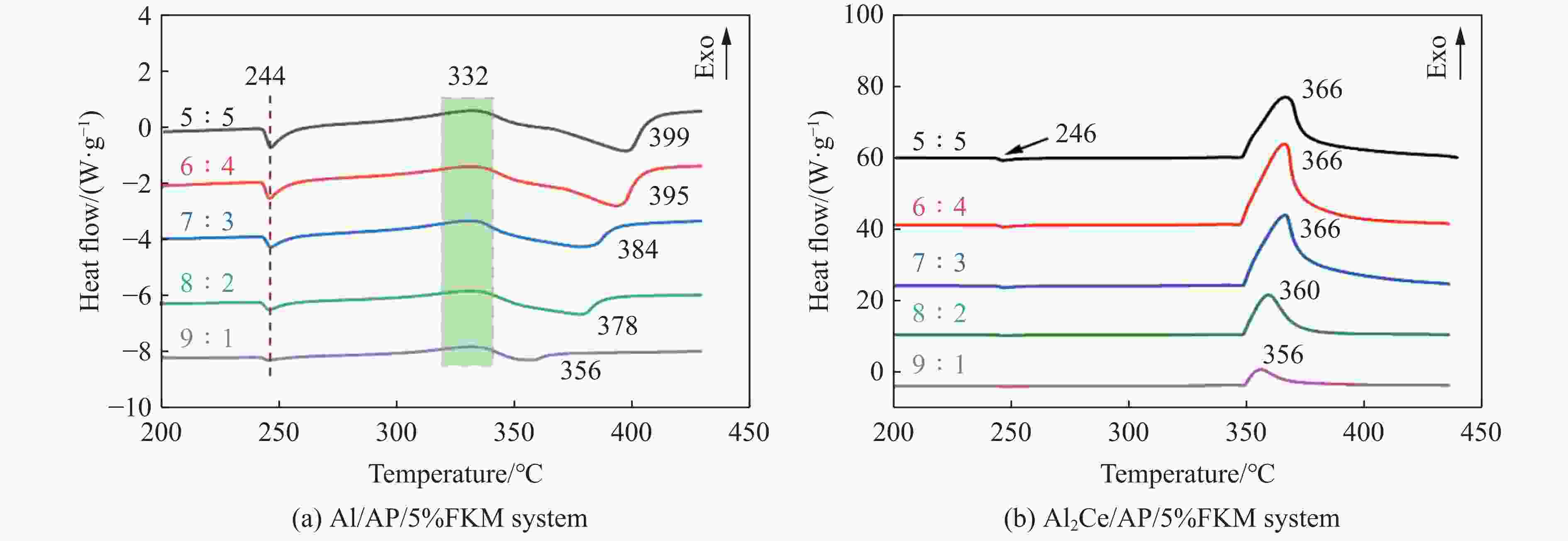
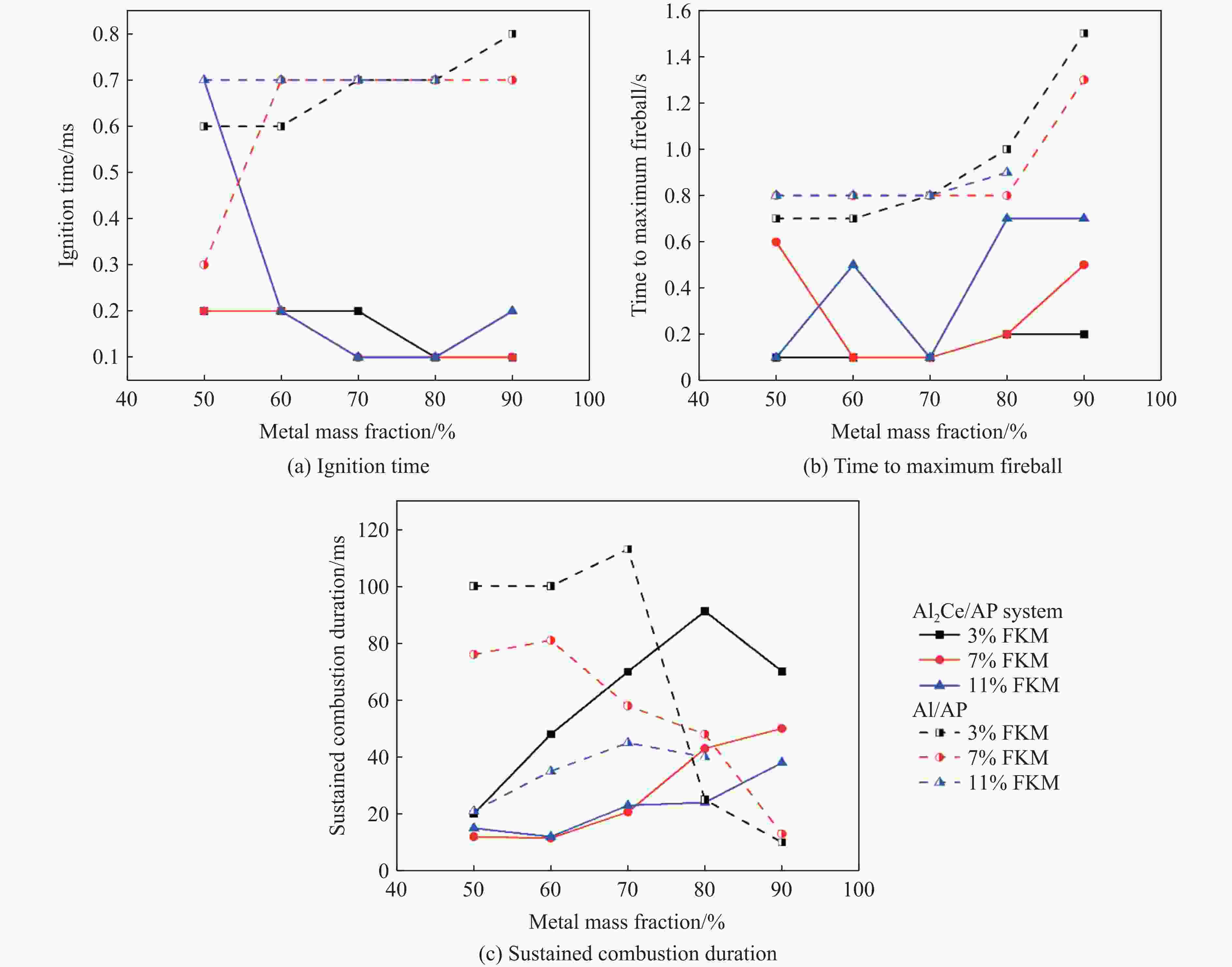
摘要: 金属铝(Al)作为常用的活性金属之一被广泛应用于活性材料体系中,但Al存在反应活性偏低制约体系能量释放的问题。为此,将铝铈合金引入反应体系中,利用稀土铈的高反应活性的特性强化铝的反应活性。为研究稀土基活性材料在冲击过载下的力学特性和点火性能,开展了Al2Ce/PTFE、Al/PTFE、Al2Ce/高氧酸铵(AP)、Al/AP 4种活性材料体系的制备和表征,通过分离式霍普金森压杆实验系统动态加载,测量了4种活性材料体系的动态应力-应变曲线、点火延迟、燃烧持续时间等性能;通过热分析测试,分析了不同含量活性金属对AP的热分解性能的影响。结果表明:4种活性材料存在未燃烧、燃烧和爆燃3种冲击点火形态;Al2Ce/PTFE和Al/PTFE材料的点火性能较差;Al2Ce/AP体系的极限强度和临界失效应变较高,且冲击点火形态为爆燃,点火延迟和持续燃烧时间均低于Al/AP体系;Ce元素的引入加速了AP的分解,并使Al2Ce/AP体系的焓值大幅提高,能量释放更集中。Ce元素可以有效地提高金属铝的反应活性,其高反应活性的特性使活性体系材料的反应进程加速,并显著强化活性材料体系在冲击作用下的能量释放。综上所述,稀土铝合金材料因具有高反应活性优势,对于研制铝基冲击反应材料具有重要意义。Abstract: Aluminum (Al), a commonly used reactive metals, is widely applied in reactive material systems. However, its relatively low reactivity restricts the energy release of systems. To improve the reactivity of aluminum, we introduced aluminum-cerium Al-Ce alloy containing the highly reactive rare earth element cerium into the system. The present study investigated the mechanical properties and ignition performance of four reactive material systems involving Al2Ce/PTFE, Al/PTFE, Al2Ce/ammonium perchlorate (AP), and Al/AP were investigated under shock overload. A split Hopkinson pressure bar (SHPB) system was used to reveal the dynamic stress-strain behavior, ignition delay, and combustion duration of the prepared samples. Thermal analysis was conducted to assess the influence of the reactive metal content on the thermal decomposition of AP. The results showed there are three distinct shock-induced ignition modes: non-ignition, combustion, and combustion (deflagration). Both Al2Ce/PTFE and Al/PTFE exhibited substandard ignition performance. The Al2Ce/AP system demonstrates higher ultimate strength and critical failure strain, achieving deflagration upon impact with significantly shorter ignition delay and combustion duration compared to Al/AP. The incorporation of the cerium accelerates AP decomposition and substantially increased the enthalpy of the Al2Ce/AP system, resulting in more concentrated energy release. Ce effectively enhances the reactivity of aluminum, and its high reactivity accelerates the reaction kinetics of the reactive system. Furthermore, it significantly intensifies energy release under impact loading. In conclusion, the rare earth aluminum alloy materials exhibit a high reactivity, which demonstrates significant potential for the development of aluminum-based impact reaction materials.
-
表 1 4种活性材料的不同体系实验样品参数
Table 1. Parameter for experimental samples of four reactive materials under different systems
Number Formulation Mass ratio FKM mass fraction/% Thickness/mm Diameter/mm 1–4 Al/PTFE 6∶4, 7∶3, 8∶2, 9∶1 4.4 ± 0.2 10.14 ± 0.02 5–9 Al2Ce/PTFE 5∶5, 6∶4, 7∶3, 8∶2, 9∶1 3.6 ± 0.2 10–34 Al2Ce/AP 5∶5, 6∶4, 7∶3, 8∶2, 9∶1 3, 5, 7, 9, 11 4.2 ± 0.2 10.16 ± 0.02 35–59 Al/AP 表 2 活性金属的基本情况
Table 2. Basic information on the reactive metals
Reactive metal Mass fraction of Al/% Crystalline phase Particle size/μm Atomized aluminum powder 100 Al 10−20 Al-Ce alloy 30 Al2Ce 75−150 表 3 实验中的主要试剂
Table 3. Main reagents for the experiment
Reagent name Purity Source NH4ClO4 Industrial grade Beifang Xing’an Chemical Co., Ltd. Polytetrafluoroethylene Analytical grade Aladdin Fluor rubber Industrial grade Zhonghao Chenguang Research Institute Co., Ltd. Petroleum ether Analytical grade Sinopharm Chemical Reagent Co., Ltd. Ethyl acetate Analytical grade Sinopharm Chemical Reagent Co., Ltd. 表 4 Al/AP(7%氟橡胶)、Al/PTFE、Al2Ce/AP(5%氟橡胶)和Al2Ce/PTFE体系的强度和临界失效应变
Table 4. Strength and critical failure strain of the Al/AP with 7% FKM, Al/PTFE system, Al2Ce/AP with 5% FKM, and Al2Ce/PTFE system
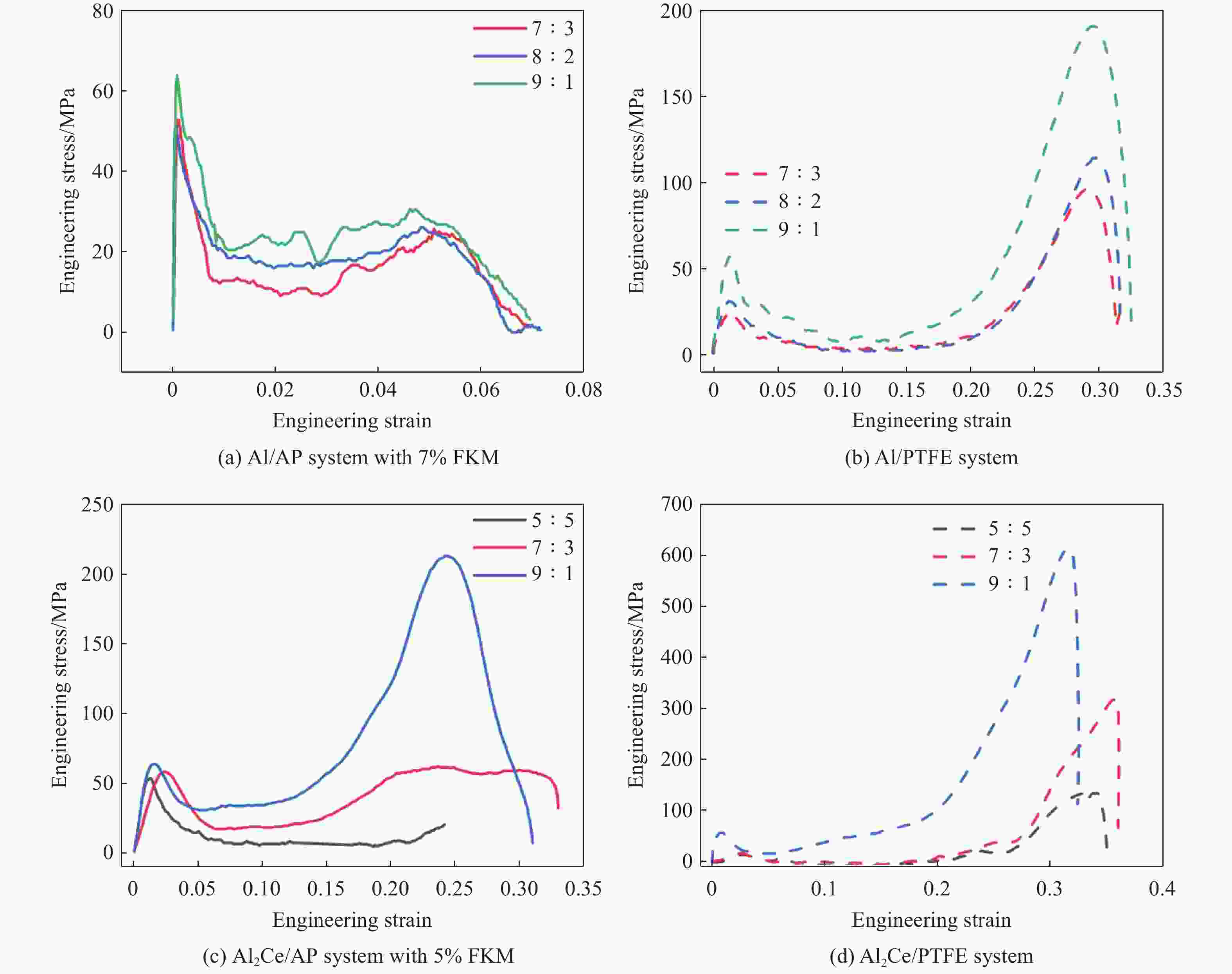
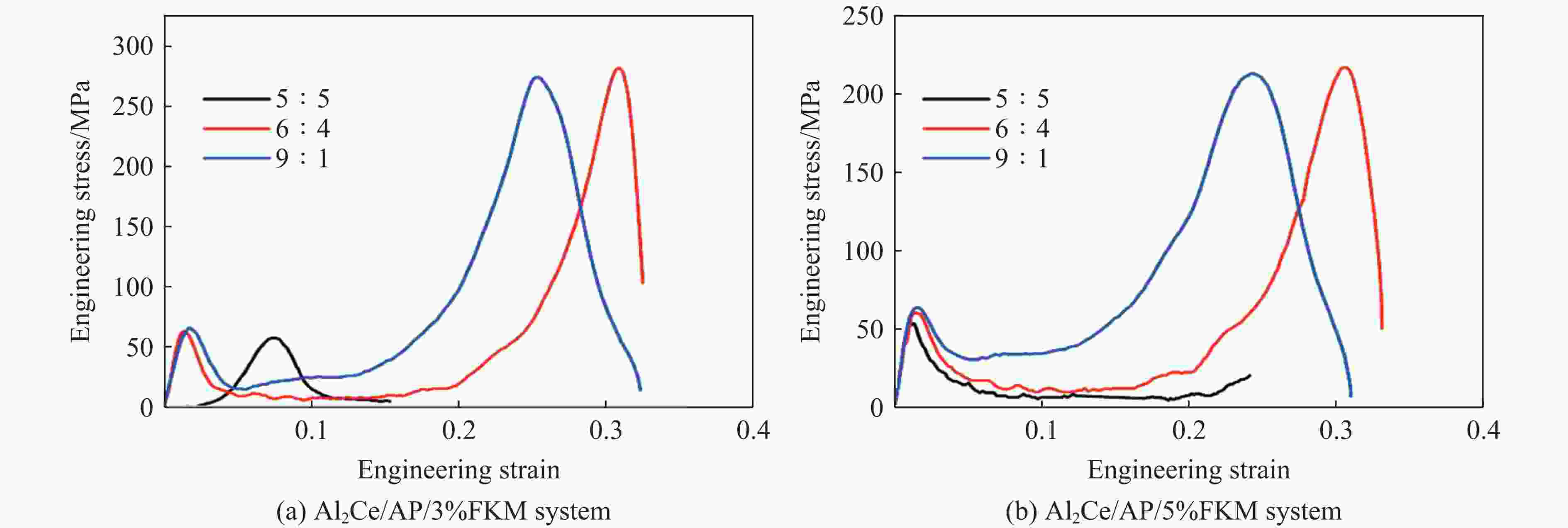
Material Mass ratio Yield strength/MPa Ultimate strength/MPa Critical failure strain Al/AP system with 7% FKM 7∶3 52.82 25.74 0.05 8∶2 51.56 26.08 0.05 9∶1 63.91 30.66 0.05 Al/PTFE system 7∶3 24.68 97.39 0.29 8∶2 31.36 114.53 0.30 9∶1 57.30 191.00 0.30 Al2Ce/AP system with 5% FKM 5∶5 53.27 7∶3 58.27 61.91 0.24 9∶1 63.56 212.32 0.25 Al2Ce/PTFE system 5∶5 13.31 132.98 0.33 7∶3 16.93 318.90 0.36 9∶1 56.44 612.35 0.32 -
[1] 汪德武, 任柯融, 江增荣, 等. 活性材料冲击释能行为研究进展 [J]. 爆炸与冲击, 2021, 41(3): 031408. doi: 10.11883/bzycj-2020-0337WANG D W, REN K R, JIANG Z R, et al. Shock-induced energy release behaviors of reactive materials [J]. Explosion and Shock Waves, 2021, 41(3): 031408. doi: 10.11883/bzycj-2020-0337 [2] MOCK W JR, DROTAR J T. Effect of aluminum particle size on the impact initiation of pressed PTFE/Al composite rods [J]. AIP Conference Proceedings, 2007, 955(1): 971–974. doi: 10.1063/1.2833292 [3] 郑雄伟, 陈进, 袁宝慧, 等. 防空反导活性破片前沿技术研究综述 [J]. 飞航导弹, 2013(9): 91–94.ZHENG X W, CHEN J, YUAN B H, et al. A review of cutting-edge technology research on air defense and anti missile active fragments [J]. Aerospace Technology, 2013(9): 91–94. [4] 叶文君, 汪涛, 鱼银虎. 氟聚物基含能反应材料研究进展 [J]. 宇航材料工艺, 2012, 42(6): 19–23. doi: 10.3969/j.issn.1007-2330.2012.06.003YE W J, WANG T, YU Y H. Research progress of fluoropolymer-matrix energetic reactive materials [J]. Aerospace Materials & Technology, 2012, 42(6): 19–23. doi: 10.3969/j.issn.1007-2330.2012.06.003 [5] GENG H H, LIU R, REN Y P, et al. Dynamic compression-shear ignition mechanism of Al/PTFE reactive materials [J]. Composite Structures, 2024, 331: 117908. doi: 10.1016/j.compstruct.2024.117908 [6] JIANG C L, HU R, ZHANG J B, et al. Shock-induced chemical reaction characteristics of PTFE-Al-Bi2O3 reactive materials [J]. Defence Technology, 2024, 36: 1–12. doi: 10.1016/j.dt.2024.01.008 [7] 赵涵, 任会兰, 宁建国. 铝纤维增强铝/聚四氟乙烯活性材料力学性能及反应特性 [J]. 兵工学报, 2024, 45(5): 1573–1581. doi: 10.12382/bgxb.2023.0101ZHAO H, REN H L, NING J G. Mechanical properties and reaction characteristics of aluminum fiber reinforced aluminum/polytetrafluoroethylene reactive material [J]. Acta Armamentarii, 2024, 45(5): 1573–1581. doi: 10.12382/bgxb.2023.0101 [8] GE C, YU Q B, ZHANG H, et al. On dynamic response and fracture-induced initiation characteristics of aluminum particle filled PTFE reactive material using hat-shaped specimens [J]. Materials & Design, 2020, 188: 108472. doi: 10.1016/j.matdes.2020.108472 [9] REN H L, LI W, NING J G, et al. The influence of initial defects on impact ignition of aluminum/polytetrafluoroethylene reactive material [J]. Advanced Engineering Materials, 2020, 22(3): 1900821. doi: 10.1002/adem.201900821 [10] XU X C, FANG H J, CHEN R, et al. Effects of different mass ratios on mechanical properties and impact energy release characteristics of Al/PTFE/W and Al/PTFE/CuO polymer composites [J]. Polymer Composites, 2024, 45(8): 7222–7236. doi: 10.1002/pc.28262 [11] YUAN Y, SHI D F, HE S, et al. Shock-induced energy release performances of PTFE/Al/oxide [J]. materials, 2022, 15(9): 3042. doi: 10.3390/ma15093042 [12] 冯彬, 方向, 李裕春, 等. 烧结温度、配比及粒径对Al-Teflon准静压反应的影响 [J]. 含能材料, 2016, 24(12): 1209–1213. doi: 10.11943/j.issn.1006-9941.2016.12.014FENG B, FANG X, LI Y C, et al. Effect of sintering temperature, ratio and particle size on the reaction of Al-teflon under quasi-static compression [J]. Chinese Journal of Energetic Materials, 2016, 24(12): 1209–1213. doi: 10.11943/j.issn.1006-9941.2016.12.014 [13] LI Y, JIANG C L, WANG Z C, et al. Experimental study on reaction characteristics of PTFE/Ti/W energetic materials under explosive loading [J]. Materials, 2016, 9(11): 936. doi: 10.3390/ma9110936 [14] 聂政元, 肖建光, 王岩鑫, 等. THV基活性材料力学性能与点火反应特性 [J]. 兵工学报, 2022, 43(12): 3030–3039. doi: 10.12382/bgxb.2021.0684NIE Z Y, XIAO J G, WANG Y X, et al. Mechanical properties and ignitionand reaction characteristics of THV-based reactive materials [J]. Acta Armamentarii, 2022, 43(12): 3030–3039. doi: 10.12382/bgxb.2021.0684 [15] 罗普光, 毛亮, 魏晨杨, 等. 锆基非晶活性材料动态力学性能及本构关系 [J]. 含能材料, 2021, 29(12): 1176–1181. doi: 10.11943/CJEM2021068LUO P G, MAO L, WEI C Y, et al. Dynamic mechanical properties and constitutive relations of Zr-based amorphous reactive material [J]. Chinese Journal of Energetic Materials, 2021, 29(12): 1176–1181. doi: 10.11943/CJEM2021068 [16] 郭连贵, 宋武林, 谢长生, 等. 核壳结构纳米铝粉热学行为 [J]. 推进技术, 2012, 33(3): 478–482. doi: 10.13675/j.cnki.tjjs.2012.03.006GUO L G, SONG W L, XIE C S, et al. Thermal behavior of core-shell structure aluminum nanopowders [J]. Journal of Propulsion Technology, 2012, 33(3): 478–482. doi: 10.13675/j.cnki.tjjs.2012.03.006 [17] GONG F Y, ZHANG J H, DING L, et al. Mussel-inspired coating of energetic crystals: a compact core-shell structure with highly enhanced thermal stability [J]. Chemical Engineering Journal, 2017, 309: 140–150. doi: 10.1016/j.cej.2016.10.020 [18] 张滨婧, 王剑. 铈及铈基材料在口腔疾病领域中的应用 [J]. 中国组织工程研究, 2024, 28(15): 2445–2451. doi: 10.12307/2024.246ZHANG B J, WANG J. Cerium and cerium-based materials in dental applications [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(15): 2445–2451. doi: 10.12307/2024.246 [19] CZERWINSKI F. Cerium in aluminum alloys [J]. Journal of Materials Science, 2020, 55(1): 24–72. doi: 10.1007/s10853-019-03892-z [20] 贾会平. 稀土金属燃烧剂的研究综述 [J]. 兵工学报, 1994(2): 70–74.JIA H P. A review of researches on mischmetal incendiary agents [J]. Acta Armamentarii, 1994(2): 70–74. [21] 吴南萍, 曾令坚, 江民涛, 等. 稀土在烟花中的应用研究 [J]. 稀土, 1989, (4): 55–56, 24.WU N P, ZENG L J, JIANG M T, et al. Research on the application of rare earth elements in fireworks [J]. Chinese Rare Earths, 1989, (4): 55–56, 24. [22] 郑世宗. 稀土对炸药能量和安定性的影响 [J]. 兵器材料科学与工程, 1988, (8): 81–84.ZHENG S Z. The influence of rare earths on the energy and stability of explosives [J]. Ordnance Material Science and Engineering, 1988, (8): 81–84. [23] 赵修臣, 关紫阳, 熊杰, 等. 氢化铈的制备及其与高氯酸铵的反应特性 [J]. 华北理工大学学报(自然科学版), 2021, 43(1): 64–70. doi: 10.3969/j.issn.2095-2716.2021.01.010ZHAO X C, GUAN Z Y, XIONG J, et al. Preparation of cerium hydride and its reaction characteristics with ammonium perchlorate [J]. Journal of North China University of Science and Technology (Natural Science Edition), 2021, 43(1): 64–70. doi: 10.3969/j.issn.2095-2716.2021.01.010 [24] 郭雨. 稀土金属复合氧化物的制备和性能研究 [D]. 西安: 西北大学, 2019: 42–49.GUO Y. Preparation and properties of rare earth metal composite oxides [D]. Xi’an: Northwest University, 2019: 42–49. [25] 刘子如, 阴翠梅, 孔扬辉, 等. 高氯酸铵的热分解 [J]. 含能材料, 2000, 8(2): 75–79. doi: 10.3969/j.issn.1006-9941.2000.02.008LIU Z R, YIN C M, KONG Y H, et al. The thermal decomposition of ammonium perchlorate [J]. Chinese Journal of Energetic Materials, 2000, 8(2): 75–79. doi: 10.3969/j.issn.1006-9941.2000.02.008 [26] ZHOU P, REN Z Q, TANG X L, et al. Interaction between Prussian blue ultrathin nanosheet and ammonium perchlorate for highly efficient thermal decomposition [J]. Advanced Functional Materials, 2023, 33(21): 2300661. doi: 10.1002/adfm.202300661 -







 下载:
下载: