High Pressure Investigation on the Alkaline-Earth Metal Hydride
-
摘要: 富氢化合物的压致金属化和超导电性是实现金属氢和高温超导体的有效途径,已成为物理学、材料科学等学科的研究热点之一。从应用上看,富氢化合物是潜在的储氢材料,研究高压下富氢化合物结构和性质变化是提升其储氢性能的有效手段。以典型的第二主族氢化物为例,简要地介绍了第二主族氢化物在高压下的实验与理论研究成果,包括高压结构相变、新结构的稳定性以及金属化机制,并探讨不同的氢构型对压致金属化及超导电性的影响。Abstract: As an effective way to realize metal hydrogen and high-temperature superconductors, the metalization and superconductivity of hydrogen-rich compounds have become one of the hot spots of physics and materials science.In practical application, hydrogen-rich compounds are also potential hydrogen storage materials.The study of the structure and properties of hydrogen-rich compounds under high pressure is considered to be an effective means to enhance their hydrogen storage performance.In this paper, high pressure experimental and theoretical researches on the second main group hydrides, a typical kind of rich hydrogen compounds, were briefly introduced, including high pressure structural phase transition, the stability of the new structure, the mechanism of metalization.The effects of different hydrogen motifs on superconductivity were also discussed.
-
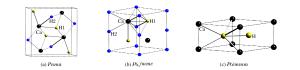
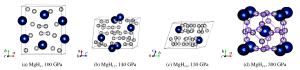
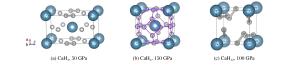
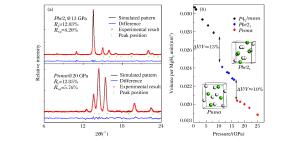
图 3 第二主族二氢化物的高压相序(实验确定的相序与实验压力范围之外的理论预测结果分别用实线和虚线标示。涉及的参考文献:[24](BeH2)、[28, 44](MgH2)、[36, 40](CaH2)、[37](SrH2)、[38-39, 41-42](BaH2)
Figure 3. High pressure phase transition sequence of alkaline earth dihydrides (The results obtained in experiments are indicated with the solid lines.The theoretical predictions beyond the experimental pressure region are indicated with dash lines.Corresponding reference:[24](BeH2), [28, 44](MgH2), [36, 40](CaH2), [37](SrH2), [38-39, 41-42](BaH2).)
-
[1] SCHLAPBACH L, ZÜTTEL A.Hydrogen-storage materials for mobile applications[J]. Nature, 2001, 414(6861):353-358. doi: 10.1038/35104634 [2] SONG Y.New perspectives on potential hydrogen storage materials using high pressure[J]. Phys Chem Chem Phys, 2013, 15(35):14524-14547. doi: 10.1039/c3cp52154k [3] HUOT J, LIANG G, BOILY S, et al.Structural study and hydrogen sorption kinetics of ball-milled magnesium hydride[J]. J Alloy Compd, 1999, 293:495-500. http://www.sciencedirect.com/science/article/pii/S0925838899004740 [4] LIANG G, HUOT J, BOILY S, et al.Catalytic effect of transition metals on hydrogen sorption in nanocrystalline ball milled MgH2-Tm (Tm=Ti, V, Mn, Fe and Ni) systems[J]. J Alloy Compd, 1999, 292(1):247-252. https://www.sciencedirect.com/science/article/abs/pii/S0925838899004429 [5] VAJEESTON P, RAVINDRAN P, KJEKSHUS A, et al.Pressure-induced structural transitions in MgH2[J]. Phys Rev Lett, 2002, 89(17):175506. doi: 10.1103/PhysRevLett.89.175506 [6] RAFIEE M A.A theoretical study of MgH2 ambient and high-pressure phases using NQCC parameters[J]. Russ J Phys Chem A, 2014, 88(13):2359-2362. doi: 10.1134/S0036024414130172 [7] WIGNER E, HUNTINGTON H B.On the possibility of a metallic modification of hydrogen[J]. J Chem Phys, 1935, 3(12):764-770. doi: 10.1063/1.1749590 [8] ASHCROFT N W.Metallic hydrogen:a high-temperature superconductor?[J]. Phys Rev Lett, 1968, 21(26):1748. doi: 10.1103/PhysRevLett.21.1748 [9] NARAYANA C, LUO H, ORLOFF J, et al.Solid hydrogen at 342 GPa:no evidence for an alkali metal[J]. Nature, 1998, 393(6680):46-49. doi: 10.1038/29949 [10] LOUBEYRE P, OCCELLI F, LETOULLEC R.Optical studies of solid hydrogen to 320 GPa and evidence for black hydrogen[J]. Nature, 2002, 416(6881):613-617. doi: 10.1038/416613a [11] EREMETS M I, TROYAN I A.Conductive dense hydrogen[J]. Nat Mater, 2011, 10(12):927-931. doi: 10.1038/nmat3175 [12] KLUG D D, YAO Y.Metallization of solid hydrogen:the challenge and possible solutions[J]. Phys Chem Chem Phys, 2011, 13(38):16999-17006. doi: 10.1039/c1cp21791g [13] MCMAHON J M, CEPERLEY D M.Ground-state structures of atomic metallic hydrogen[J]. Phys Rev Lett, 2011, 106(16):165302. doi: 10.1103/PhysRevLett.106.165302 [14] ZHA C S, LIU Z, HEMLEY R J.Synchrotron infrared measurements of dense hydrogen to 360 GPa[J]. Phys Rev Lett, 2012, 108(14):146402. doi: 10.1103/PhysRevLett.108.146402 [15] EREMETS M I, TROYAN I A, LERCH P, et al.Infrared study of hydrogen up to 310 GPa at room temperature[J]. High Pressure Res, 2013, 33(2):377-380. doi: 10.1080/08957959.2013.794229 [16] DIAS R P, SILVERA I F.Observation of the Wigner-Huntington transition to metallic hydrogen[J]. Science, 2017, 355(6326):715-718. doi: 10.1126/science.aal1579 [17] ASHCROFT N W.Hydrogen dominant metallic alloys:high temperature superconductors?[J]. Phys Rev Lett, 2004, 92(18):187002. doi: 10.1103/PhysRevLett.92.187002 [18] DROZDOV A P, EREMETS M I, TROYAN I A, et al.Conventional superconductivity at 203 kelvin at high pressures in the sulfur hydride system[J]. Nature, 2015, 525(7567):73-76. doi: 10.1038/nature14964 [19] DUAN D F, LIU Y X, TIAN F B, et al.Pressure-induced metallization of dense (H2S)2H2 with high-Tc superconductivity[J]. Sci Rep, 2014, 4:6968. doi: 10.1038/srep06968 [20] DUAN D F, HUANG X L, TIAN F B, et al.Pressure-induced decomposition of solid hydrogen sulfide[J]. Phys Rev B, 2015, 91(18):180502. doi: 10.1103/PhysRevB.91.180502 [21] WANG H, JOHN S T, TANAKA K, et al.Superconductive sodalite-like clathrate calcium hydride at high pressures[J]. Proc Natl Acad Sci, 2012, 109(17):6463-6466. doi: 10.1073/pnas.1118168109 [22] ARMSTRONG D R, JAMIESON J, PERKINS P G.The electronic structures of polymeric beryllium hydride and polymeric boron hydride[J]. Theoretica Chimica Acta, 1979, 51(2):163-172. doi: 10.1007/BF00554099 [23] SMITH G S, JOHNSON Q C, SMITH D K, et al.The crystal and molecular structure of beryllium hydride[J]. Solid State Commun, 1988, 67(5):491-494. doi: 10.1016/0038-1098(84)90168-6 [24] VAJEESTON P, RAVINDRAN P, KJEKSHUS A, et al.Structural stability of BeH2 at high pressures[J]. Appl Physics Lett, 2004, 84(1):34-36. doi: 10.1063/1.1637967 [25] AHART M, YARGER J L, LANTZKY K M, et al.High-pressure Brillouin scattering of amorphous BeH2[J]. J Chem Phys, 2006, 124(1):014502. doi: 10.1063/1.2138692 [26] BORTZ M, BERTHEVILLE B, BÖTTGER G, et al.Structure of the high pressure phase γ-MgH2 by neutron powder diffraction[J]. J Alloy Compd, 1999, 287(1/2):L4-L6. http://www.sciencedirect.com/science/article/pii/S0925838899000286 [27] VAJEESTON P, RAVINDRAN P, HAUBACK B C, et al.Structural stability and pressure-induced phase transitions in MgH2[J]. Phys Rev B, 2006, 73(22):224102. doi: 10.1103/PhysRevB.73.224102 [28] MORIWAKI T, AKAHAMA Y, KAWAMURA H, et al.Structural phase transition of rutile-type MgH2 at high pressures[J]. J Phy Soc Jpn, 2006, 75(7):074603. doi: 10.1143/JPSJ.75.074603 [29] ZHANG L J, WANG Y C, CUI T, et al.CaCl2-type high-pressure phase of magnesium hydride predicted by ab initio phonon calculations[J]. Phys Rev B, 2007, 75(14):144109. doi: 10.1103/PhysRevB.75.144109 [30] CUI S X, FENG W X, HU H Q, et al.Structural phase transitions in MgH2 under high pressure[J]. Solid State Commun, 2008, 148(9):403-405. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=941c8a76e49dddec2d79e92804889616 [31] ZINTL E, HARDER A.Constitution of alkaline earth hydrides[J]. Z Elektrochem, 1935, 41:33. [32] LEGER J M, HAINES J, ATOUF A.The high pressure behaviour of the cotunnite and post-cotunnite phases of PbCl2 and SnCl2[J]. J Phys Chem Solids, 1996, 57(1):7-16. http://www.sciencedirect.com/science/article/pii/0022369795000607 [33] LEGER J M, HAINES J, ATOUF A, et al.High-pressure X-ray and neutron-diffraction studies of BaF2:an example of a coordination number of 11 in AX2 compounds[J]. Phys Rev B, 1995, 52(18):13247. doi: 10.1103/PhysRevB.52.13247 [34] OLSEN J S, BURAS B, GERWARD L, et al.A new high-pressure phase and the equation of state of YbH2[J]. Physica Scripta, 1984, 29(5):503. doi: 10.1088/0031-8949/29/5/016 [35] LI B, LI Y W, YANG K F, et al.Raman evidence of a new high-pressure phase in calcium hydride[J]. J Phys Condens Matter, 2007, 19(22):226205. doi: 10.1088/0953-8984/19/22/226205 [36] TSE J S, KLUG D D, DESGRENIERS S, et al.Structural phase transition in CaH2 at high pressures[J]. Phys Rev B, 2007, 75(13):134108. doi: 10.1103/PhysRevB.75.134108 [37] SMITH J S, DESGRENIERS S, KLUG D D, et al.High-density strontium hydride:an experimental and theoretical study[J]. Solid State Commun, 2009, 149(21):830-834. http://d.old.wanfangdata.com.cn/NSTLQK/NSTL_QKJJ0210981365/ [38] SMITH J S, DESGRENIERS S, TSE J S, et al.High-pressure phase transition observed in barium hydride[J]. J Appl Phys, 2007, 102(4):043520. doi: 10.1063/1.2772427 [39] KINOSHITA K, NISHIMURA M, AKAHAMA Y, et al.Pressure-induced phase transition of BaH2:post Ni2 in phase[J]. Solid State Commun, 2007, 141(2):69-72. doi: 10.1016/j.ssc.2006.09.045 [40] LI Y W, LI B, CUI T, et al.High-pressure phase transformations in CaH2[J]. J Phys Condens Matter, 2008, 20(4):045211. doi: 10.1088/0953-8984/20/04/045211 [41] LUO W, AHUJA R.Ab initio prediction of high-pressure structural phase transition in BaH2[J]. J Alloy Compd, 2007, 446:405-408. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=95289a20a20650e1676663509f329533 [42] CHEN C B, TIAN F B, WANG L C, et al.New high-pressure phase of BaH2 predicted by ab initio studies[J]. J Phys Condens Matter, 2010, 22(22):225401. doi: 10.1088/0953-8984/22/22/225401 [43] ZHANG C, CHEN X J, ZHANG R Q, et al.Chemical trend of pressure-induced metallization in alkaline earth hydrides[J]. J Phys Chem C, 2010, 114(34):14614-14617. doi: 10.1021/jp103968c [44] LONIE D C, HOOPER J, ALTINTAS B, et al.Metallization of magnesium polyhydrides under pressure[J]. Phys Rev B, 2013, 87(5):054107. doi: 10.1103/PhysRevB.87.054107 [45] ZUREK E, HOFFMANN R, ASHCROFT N W, et al.A little bit of lithium does a lot for hydrogen[J]. Proc Natl Acad Sci, 2009, 106(42):17640-17643. doi: 10.1073/pnas.0908262106 [46] SZCZESNIAK R, DURAJSKI A P.Superconductivity well above room temperature in compressed MgH6[J]. Front Phys, 2016, 11(6):117406. doi: 10.1007/s11467-016-0578-1 [47] HOOPER J, ALTINTAS B, SHAMP A, et al.Polyhydrides of the alkaline earth metals:a look at the extremes under pressure[J]. J Phys Chem C, 2013, 117(6):2982-2992. doi: 10.1021/jp311571n [48] FENG X L, ZHANG J R, GAO G Y, et al.Compressed sodalite-like MgH6 as a potential high-temperature superconductor[J]. RSC Adv, 2015, 5(73):59292-59296. doi: 10.1039/C5RA11459D [49] HOOPER J, TERPSTRA T, SHAMP A, et al.Composition and constitution of compressed strontium polyhydrides[J]. J Phys Chem C, 2014, 118(12):6433-6447. doi: 10.1021/jp4125342 [50] PÉPIN C, LOUBEYRE P, OCCELLI F, et al.Synthesis of lithium polyhydrides above 130 GPa at 300 K[J]. Proc Natl Acad Sci, 2015, 112(25):7673-7676. doi: 10.1073/pnas.1507508112 [51] KUNO K, MATSUOKA T, NAKAGAWA T, et al.Heating of Li in hydrogen:possible synthesis of LiHx[J]. High Pressure Res, 2015, 35(1):16-21. doi: 10.1080/08957959.2014.999677 [52] STRUZHKIN V V, KIM D Y, STAVROU E, et al.Synthesis of sodium polyhydrides at high pressures[J]. Nat Commun, 2016, 7:12267. doi: 10.1038/ncomms12267 -






 下载:
下载: